Despite being one of our key senses, the molecular mechanisms of smell remain poorly understood. Mason Wakley sniffs out the competing theories of what may be happening in our noses
-
Scientists still don’t fully understand how smell works, despite it being a fundamental sense; multiple competing theories exist, and none can comprehensively explain how humans detect and distinguish odors.
-
The traditional shape (lock-and-key) theory suggests odorant molecules bind to receptors based on their structure, but inconsistencies – such as similarly shaped molecules smelling different – limit its explanatory power.
-
The vibrational theory proposes that smell depends on molecular vibrations detected via electron transfer mechanisms, but it also faces challenges, including difficulty explaining why some structurally identical molecules (like enantiomers) smell different.
-
New research indicates that smell likely involves a combination of factors – including receptor shape, molecular properties, mixtures of chemicals and possibly breakdown products – supported by advances in receptor imaging and computation, though a complete model remains elusive.
This summary was generated by AI and checked by a human editor
Imagine that you’re walking through a forest. There’s the soft crunch of the earth beneath your feet and the warmth of the sunbeams that find their way through the leaves above. A gentle breeze lifts the faint aroma of pine needles, a whole host of floral scents – and the gut-wrenching stink of an ogre standing nearby. You are, after all, in a virtual reality (VR) experience.
Introducing ‘virtual scents’ to computer simulations can make them feel more lifelike, with companies such as Scentient in the US beginning to bring this technology to market. A wraparound device sits underneath the VR headset, releasing specific fragrances depending on what users see.
Perfume companies are also looking to enter the world of virtual scents to expand the way that they advertise new fragrances to consumers. Certain start-ups are even looking at ways to turn smells into digital code to design even better smelling scents or improve the ability of sensors to detect specific signals.
But while scientists are making strides at bringing scents to the virtual world, how this fundamental sense works remains poorly understood. There are several different theories about how our nose detects and registers scents, though scientists can’t settle on which one, if any, is best.
How the shape theory explains smell
The simplest way of explaining the sense of smell is with the classic ‘lock and key’ model, first suggested by Emil Fischer in the 1890s to describe how substrates interact with enzymes. Building on this idea, the Scottish chemist Robert Moncrieff proposed the same model in the 1940s to explain how odorant molecules bind to receptors in the upper nasal cavity.
Shape theory fits well with the observation that molecules with the same functional group tend to smell similar. For example, esters smell fruity, sulfur compounds stink of eggs and cyanide molecules leave a room smelling like bitter almonds (a room that you should leave very quickly, I might add).
These binding events then send a signal to the olfactory bulb, a small organ that sits beneath the brain, which processes the signals before sending them to other regions of the brain. This includes the hippocampus, amygdala and orbitofrontal cortex, which deal with emotions and store memories, explaining why a whiff of a forgotten scent can unlock a hidden feeling.
As humans can distinguish over 10,000 different smells, chemists thought that it was unlikely that the human nose has the same number of different receptors, each uniquely adapted for different chemical shapes. As a result, UK chemist John Amoore refined Moncrieff’s idea, proposing in 1963 that there were only seven primary odours – camphoraceous, musky, floral, peppermint, ethereal, pungent and putrid – that bind to different receptors in the nose. Most scents, he thought, came from a combination of these primary odours in varying ratios.
While the lock and key model may have worked well for substrates in enzymes, however, it worked less well to explain the scent of all molecules. For example, molecules that are nearly identical in shape can smell wildly different, such as the stereoisomers R - and S -carvone that smell like spearmint and caraway seeds, respectively. Equally, while the hydrocarbon camphane smells like earthy camphor, decaborane – with a very similar structure but where boron atoms replace carbons – smells like eggy sulfur.
Yet compounds with vastly different shapes can smell distinctly similar. Muscone, musk xylene, galaxolide and helvetolide, for example, all carry the same warm and sweet musk scent and activate the same odorant receptor. The lack of a clear structure–odour relationship left some of the scientific community questioning the simplicity of this model, and looking elsewhere for an explanation of this sense.
Vibrational theory offers an alternative
As an alternative to shape theory, some scientists suggested that receptors in our nose detected subtle molecular movements to distinguish between scents.
‘Vibrational theory is a very old idea,’ says Luca Turin, who recently left the University of Buckingham in the UK to start an R&D company spinb.io. He has been a key advocate for the theory since the 1990s and explains that the idea of vibrations triggering scent dates back to the late 1920s. In those days, the main way to identify and study molecules was by using an infrared (IR) spectrometer, with nuclear magnetic resonance not developed until after the second world war.
Turin explains that chemists noticed two things when looking at IR spectra. ‘One was that the fingerprint region of the [IR] spectrum, which is between 500 and 1500cm–1, is absolutely different for each molecule.’ He adds that above 1500cm–1 individual functional groups – such as alcohols, alkynes, carbonyls and nitriles – have distinct signals.
‘[Chemists] have always described the smell of molecules in terms of their functional groups that are attached to them,’ says Turin. ‘It doesn’t matter what the functional group is attached to, in the sense that if you take a lemony skeleton [backbone] and you put a nitrile on, it [smells like] metallic lemon.’
Likening the nose to an IR spectrometer, UK chemist George Dyson suggested in 1928 that the nose was smelling molecular vibrations, rather than distinguishing between the 3D shape of molecules. Robert Wright later championed this idea in the early 1960s, helping to keep the idea alive.
Unfortunately, explains Turin, chemists at the time had limited ways to test these theories. ‘And there was another wrinkle, which was that most vibrations above about 600cm–1 are not going to be thermally excited [at room temperature],’ he adds. This means that our noses would be unable to smell certain functional groups like nitriles or alcohols. ‘So this eventually caused a kind of gradual loss of interest in vibrational theories,’ says Turin.
GPCR receptors enter the smell story
In 1991, the discovery by Richard Axel and Linda Buck of a collection of genes that code for odorant receptors sparked a resurgence in scientists trying to understand how we smell. The pair used rats to track how odours travelled from cells in the nose to the brain, and cloned several of the proteins, revealing that they were in fact G protein-coupled receptors (GPCRs). This discovery would lead them to win the 2004 Nobel prize in physiology or medicine.
Axel and Buck found that rats have around 1000 different receptors, while humans, as later studies revealed, have around 400 active odorant receptors, making up approximately 2–5% of the human genome – the largest gene family in humans.
Shape theory began to resurface on the back of their discovery. For molecules to activate GPCRs, scientists think that molecules need to have a specific conformation or shape. However, ‘there’s no direct computational evidence that you can turn on a GPCR simply by binding,’ says Turin. As a result, in the mid-1990s, Turin and others began resurrecting and advocating for vibrational theory.
In one of Turin’s first experiments, he used the idea of inelastic electron tunnelling to rationalise how molecular vibrations generated smell. He suggested that receptor proteins initially accept electrons from a biological electron donor such as the reductant NADPH (nicotinamide adenine dinucleotide phosphate). Then, when an odorant molecule enters the receptor, the electrons in the receptor can lose energy by tunnelling through the molecule, exciting the molecule’s vibrational modes. These electrons then have varying amounts of energy, which as they flow through the rest of the protein, generates a unique signal for each molecule.
‘It is very hard to imagine that [odorant receptors] would have invented a completely different mechanism for receptor activation than the rest of the GPCRs,’ says Turin. ‘My personal belief, which I reached embarrassingly slowly over the years, is that if olfactory receptors work by an electronic mechanism, then that electronic mechanism in a slightly different version, with or without vibrations, must apply to all GPCRs,’ says Turin. He adds that if electrons power GPCRs, this would alter how chemists design drugs, instead tuning electronic properties rather than binding affinity to increase drug efficacy.
Testing smell theories with isotopes
Differences in smell between isotopic analogues – molecules with the same structure but with heavier deuterium atoms replacing hydrogens – became a key way to probe vibrational theory. Vibrational frequencies are inversely proportional to the reduced mass of the atoms in a bond, meaning bonds containing deuterium vibrate at lower frequencies and therefore should smell different.
Early studies found that insects and fish were able to tell deuterated and non-deuterated molecules apart. Equally, vibrational theory helped explain why certain borane compounds smelled sulfurous, despite containing no sulfur atoms, as the boron–hydrogen and sulfur–hydrogen bond vibrations are similar in frequency (~2600 and ~2500cm–1, respectively). Turin explains that the number of hydrogens replaced by deuterium may also influence how different the molecules smell, and the distinction may not be possible in small molecules with few carbon–hydrogen or carbon–deuterium bonds.
In 2001, scientists also claimed that humans were able to tell benzaldehyde apart from its deuterated form, a molecule with only three carbon–hydrogen bonds. However, others have since rejected the idea as there was no double-blind trial as part of the analysis. Other evidence against vibrational theory soon started to emerge. ‘The vibration theory of olfaction cannot explain why we can discriminate enantiomers because they do have the exact same vibrations,’ says Hiroaki Matsunami, a geneticist who studies olfaction at Duke University School of Medicine in North Carolina, US.
Previous studies had suggested that humans could tell the difference between isotopic analogues of the musk compound cyclopentadecanone, but later analysis with human and mouse odorant receptors challenged this idea, as these proteins responded similarly to normal, deuterated and carbon-13 isotopic analogues.
Cryo-EM reveals receptors in action
As shape and vibrational theory both cannot not fully explain our sense of smell, scientists began probing this sense in alternative ways. One way to do that was to study the 3D structure of odorant receptors with bound scent molecules. Such receptors come in two types: Class I or ‘carboxylic acid’ receptors that detect water-soluble odorants, and Class II receptors that smell everything else, mostly hydrophobic compounds.
Matsunami explains that to determine the structure of typical receptor proteins, the standard route would be to isolate and purify the protein, and then analyse the crystal structure. However, he adds that this method is a challenge with odorant receptors as they are among the most difficult proteins to work with.
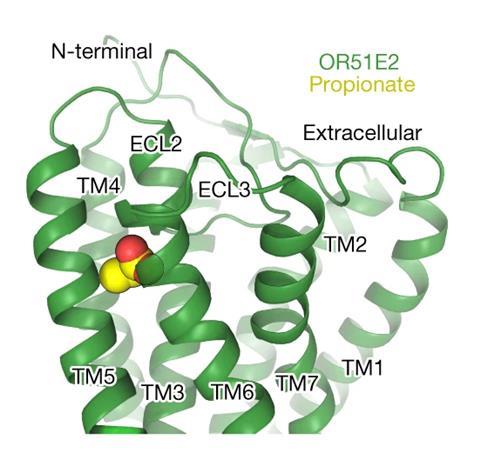
Progress with cryo-electron microscopy (cryo-EM) in recent years has helped overcome this issue. To get an accurate signal, scientists now only need around 10µg of protein sample, instead of several milligrams, explains Matsunami. As a result, in 2023, Matsunami and his team was able to use cryo-EM to capture how the carboxylic acid propionate interacts with the human odorant receptor, OR51E2, a class I receptor. This marked a first for the field.
The team chose the OR51E2 receptor as it is a well-expressed protein, compared to other odorant receptors, making it easier to generate the amount of sample required. After analysing the structure of the receptor, the team suggested that when propionate binds, one of the transmembrane helices that make up the tertiary structure of the protein rotates inward, activating the receptor. Propionate may also bind with both ionic and hydrogen bond interactions that help anchor the carboxylic acid in place, as well as less specific hydrophobic interactions that rely on the molecule’s shape. The changes in shape may generate a signal, with the team suggesting that this binding mechanism may apply to other class I receptors that recognise polar, water-soluble molecules.
‘The current way of thinking is that it’s not only the shape, but also other characteristics of both odorants and the binding cavity of the receptors,’ explains Matsunami. He adds that GPCRs can take on multiple active and inactive confirmations, and while a receptor may be very receptive to a particular molecule, a high concentration of a weakly binding odorant may also trigger a response.
‘It’s great to do this experimental approach, but it is not feasible to do it for all 400 receptors … and therefore a computational approach is really, really powerful,’ explains Matsunami. He adds that modelling tools such as AlphaFold – which won the Nobel prize in chemistry in 2024 – may help aid in-lab experiments to analyse hundreds of receptors at once to better understand the whole picture of smell.
The future of smell
Experiments new and old show that the situation of how our noses detect scent is more complicated than simply the shape or vibrations of molecules. Others are now taking different angles to figure out how this sense works. Students in California, US, have recently demonstrated that they are able to stimulate the olfactory bulb by pointing ultrasound waves at the organ through the skull. Ultrasound waves have a frequency greater than 20kHz, which is above the human level of hearing. Turin stresses that this experiment is ‘a reckless enterprise’ as ‘putting reasonably high intensity ultrasound onto the brain tissue is not a good idea’.
However, if done properly, he thinks that it is an interesting concept. ‘The sense of smell is the only sense where no one has been able to elicit a sensation by peripheral stimulation,’ he says. ‘[The students] find that when they go back and forth [with the ultrasound], they get reliably different sensations – they get smoky at one point and fresh air at another.’ The students suggest that this method could allow scientists to encode information in ultrasound frequencies and then send these to users as a way to learn information through smell, likened to the way that certain people can hear colours.
‘This is tonotopic organisation, just like in the ear,’ Turin says, explaining that the brain processes sound frequencies in different regions. ‘This is probably the most important discovery in smell in the last 10–15 years,’ he adds, although the results have yet to be peer reviewed or carried out with a large sample of people.
On the other hand, Matsunami hypothesises that what we smell is not necessarily the odorant molecule itself, but instead the products of a molecule breaking down. ‘For example, esters can by hydrolysed into different molecules and then these metabolites bind to the receptor, and maybe that’s going to affect your sense.’ He rationalises that deuterium may affect these types of reactions, which may have some role in explaining the observation that isotopic analogues can smell different.
He also stresses that ‘most odours we encounter in everyday life are a complex mixture’. ‘If you smell bread or coffee, they’re basically coming [to you] as a mixture of probably hundreds of different kinds of chemicals.’ Scientists have just started to probe how a single receptor with a single molecule works, but understanding the ‘mixture interaction at the receptor level is a very important question to be answered’, Matsunami says.
While figuring out how this fundamental sense works is exciting, he thinks that this knowledge will also have a more practical use. ‘If you start understanding the pharmacology of odorant receptors, then you can now use, for example, computational approaches to design a new molecule that may smell even better,’ he adds.
‘There are [also] many different kinds of chemical sensors around but none of them are as good as trained dogs,’ he says, explaining that these animals are capable of detecting drugs, diseases and explosive materials, even at very low concentrations. Completing our understanding of smell may lead to better physical detectors, which he thinks is a ‘quite useful thing’.
It’s clear that fully understanding how receptors in our nose detect molecules still has a long way to go, with a combination of shape, vibrational structure and metabolites perhaps creating a unique signal for each specific smell. So, the next time you stop to smell the roses – be that in real life or in a VR setting – take a pause to think about the complex chemistry happening right beneath our noses.
Mason Wakley is a science correspondent for Chemistry World



















No comments yet