From crystallographers’ beards to billion-dollar lawsuits, the phenomenon of disappearing polymorphs has puzzled chemists for decades. Now researchers are finally unravelling the science behind why stable crystal forms suddenly vanish – and discovering how to bring them back
- Disappearing polymorphs occur when a new, more stable crystal form of a compound suddenly emerges and permanently replaces the original form, often due to subtle shifts in nucleation conditions or accidental environmental ‘seeding’.
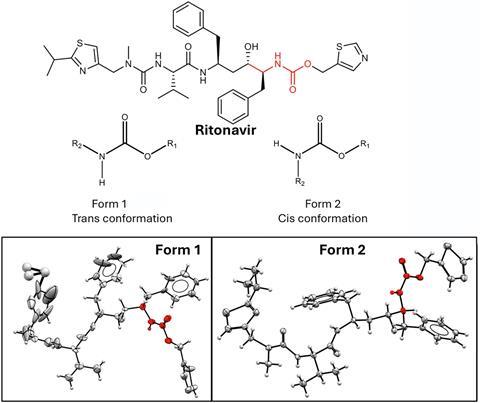
- This phenomenon has caused major pharmaceutical disruptions, most famously with ritonavir, where the unexpected appearance of a new polymorph rendered the original drug formulation ineffective and forced costly reformulation.
- Disappearing polymorphs have also played a key role in high‑stakes patent battles, with companies arguing over whether new crystal forms were truly novel or inevitably produced by existing methods.
- Advances in crystal engineering and modelling — including mechanochemical methods that can interconvert polymorphs and computational tools predicting nucleation behaviour — are beginning to offer explanations and potential control over this once‑mysterious phenomenon.
This summary was generated by AI and checked by a human editor
One of chemistry’s best loved ‘urban myths’, the crystallographer’s beard can be traced to Gerhard Schmidt, the German-born scientific director of the Weizmann Institute of Science in the 1960s. He recounted that when a student was unable to crystallise a product, the great 19th century German chemists Baeyer and Wallach would come and shake their beard over the flask to deposit a stray crystal, instantly seeding crystallisation. He suggested the modern beardless chemist create an equivalent device with a salt-shaker of differently shaped microcrystals.
Schmidt was forced to escape Nazi Germany at 16 so the story is unlikely to have been passed on from an actual student of one of these great chemists, but the chemistry behind it is more than just a crystallographer’s in-joke. The idea that crystallisation can be controlled by seeding with micro crystals is well-established – although generally not from a beard. Seeding is also partly responsible for the curious phenomenon of the disappearing polymorph, where the dominant crystal form of a molecule disappears and is superseded by a new form.
Disappearing polymorphs have caused real problems for pharma at times, but the concept has also been used as a legal device to keep generic drug-makers at bay. Today, with an ever-increasing understanding of the chemistry behind crystallisation, the phenomenon may soon become a curious footnote or another chemistry urban myth.
From myth to reality
A well-known example of the disappearing polymorph is the HIV/Aids drug ritonavir, developed by Abbott Laboratories, which gained approval in 1996. Two years later a more stable, less soluble crystalline form (form II) began appearing in manufactured batches, replacing the original form I and rendering the capsules ineffective. ‘The solubility was so low that the drug had difficulties acting in the body because the level of the [active pharmaceutical ingredient] in the blood was not high [enough],’ explains Joop der Horst, a process scientist and polymorph expert at pharmaceutical manufacturer Tiofarma, based in the Netherlands. Abbott was forced to temporarily withdraw the drug until a new more stable refrigerated gel-capsule was produced that contained the original polymorph. The episode cost the firm over $250 million.
It was not clear why the new, more stable form suddenly appeared, explains Aurora Cruz-Cabeza, a chemist from the University of Durham in the UK, but ‘once you got small crystal seeds present, then it was really easy to grow and everything converted to the new polymorph, and [Abbott] couldn’t go back to producing the original crystal form.’
Growing crystals has always been half science and half art, and over the years has attracted some left-field theories. In the 1980s for example, the biochemist turned parapsychologist Rupert Sheldrake said his ‘morphic resonance’ theory, that nature has a kind of collective memory, explained why compounds become easier to crystallise over time.
But the real explanation comes down to some of the basic and very well-established principles of chemical theory. The battle between polymorphs is a balancing act between thermodynamics and kinetics, which means the first crystals that form can be a less thermodynamically stable phase, if it forms more quickly. ‘Often you see that the kinetics of formation of the metastable form is faster than the stable form,’ says der Horst, ‘it somehow has the ability to more easily pack together.’ Even though one polymorph may be thermodynamcially more stable, it can be handicapped by the nucleation process – how those first molecules in solution come together and ultimately form a sold phase.
The speed of nucleation is controlled by the surface energy of the emerging molecular clusters, says materials scientist Wenhao Sun from the University of Michigan in the US, who has been modelling the nucleation process. ‘When crystals are small, they have a lot of surface area relative to their volume,’ he says. The high surface energy – the energy required to form a surface – tends to act against crystal formation with very small clusters of molecules falling apart rather than continuining to grow, known as the nucleation barrier. At a certain size, however, the clusters overcome the barrier. ‘Once it overcomes this hill, then it can form a crystal and grow continuously,’ explains Sun. So if a metastable phase has a lower surface area and hence a lower nucleation barrier than the more thermodynamically stable polymorph, it tends to form first and then keeps forming.
Sun says when you look at a picture of the molecular structures of the two ritonavir polymorphs you can almost see the difference in surface energies without even having to do calculations. ‘Form II is denser, it’s more interconnected, there are more hydrogen bonds … it also feels like higher surface energy, which means its harder to cleave.’ All this means that form II would be less likely to nucleate.
But he adds that if form II ‘were to nucleate, the whole system would transform from form I to form II’, which is what ultimately happened. Why it happened after two years for ritonavir is still unclear. Der Horst says the balance between polymorphs ‘is easily influenced by external parameters’. One suggestion is a change in synthesis method could have led to different byproducts, or impurities from a new source of raw materials or a change in solvent. Anything that slows down the nucleation of the metastable form could end up tipping the balance to the new polymorph.
But it could just be a random, stochastic event, says Sun. ‘Particles coming together, and then if they overcome [the energy barrier] enough, then you can transform to this lower energy phase.’ This might be a rare event, ‘one in every billion or one in every trillion [tries]’ says Sun – or once every two years. ‘Once you start growing the more stable form, that stable form can shed crystals, and when it sheds crystals, that gives you more seeds, and then you can never recover the old, higher energy, metastable phase.’ he adds.
When polymorphs cost millions
There is still the mystery of why once the stable form is produced the metastable polymorph seems to quickly disappear almost everywhere. This leads back to the idea of the crystallographer’s beard. Chemical industry consultant Derek Robinson, now based in Scotland, recounts the story that a Takeda Pharmaceuticals research chemist, Mitsuhisa Yamano, presented at a polymorphism conference in 2007. During development of Takeda’s broad spectrum cephalosporin antibiotic cefozopran in 1986, the company readily obtained the alpha polymorph, a crystalline trihydrate. Then four years later a new less-soluble but more stable gamma crystal appeared, a heptahydrate. The alpha-form was never obtained again at any of Takeda’s labs.
Robinson points out that these were not strictly polymorphs as they had a different number of water molecules in the crystals, but the principle and manner of the disappearance was the same. Yamano had carefully documented how the new polymorph first turned up in an analysis lab, and the following day in the process research building, then one day later in the pilot plant, and within a few weeks all over the campus. He concluded that microscopic crystals of the new form had become present in the environment, presumably travelling with research staff or equipment, gradually seeding the new polymorph everywhere. It even quickly turned up at Takeda’s drug safety research lab, located 12 miles away.
It seems far-fetched but ‘why not?’ asks Roger Davey, an expert in crystal nucleation and polymorphism, now retired from the University of Manchester, UK. ‘There’s dust everywhere – I wouldn’t write it off.’
The disappearing polymorph idea has also been at the centre of several high stakes US patent cases from the 1990s to the early 2000s. The first was related to ranitidine hydrochloride, marketed by Glaxo (now GSK) as Zantac to treat peptic ulcers. Patent attorney Thorsten Bausch, now a partner at Hoffmann Eitle in Germany, was a young associate at the time. ‘It was a significant case, first of all, because so much money was at stake,’ he recalls
Glaxo had patented the polymorph form I but when a second, form II, appeared, it completely replaced the first, despite the method for its production remaining the same. Glaxo then patented form II, but when the first patent expired in 1993, Canadian company Novopharm sought approval from the US courts to produce a generic version of the new polymorph. They argued that because the method to make form I now always resulted in form II, the new patent on this polymorph has been ‘inherently anticipated’ in the original patent. Bausch explains this legal concept means that an invention is not considered new if an existing method inevitably leads to it, in this case the new polymorph. It would mean the second patent would be invalid.
But Glaxo countered that the method did not inevitably lead to form II and the original polymorph could still be produced in a seed-free clean environment. They employed two Oxford organic chemistry postdocs to show it could be done on three separate occasions. Bausch says the ‘flat out contradictory evidence’ of both sides could be explained by the disappearing polymorph phenomena, but ultimately the court decided this wasn’t enough to invalidate the patent, which would have held up at the time form I was first produced, before the new polymorph arose.
Novopharm went back to the lab and produced its own version of the older (now out of patent) form I of ranitidine hydrochloride. But Glaxo again sued, now claiming that because of the disappearing polymorph phenomenon, any attempt to make form I would now include some traces of the more stable form II, infringing its second patent. Glaxo was unable to prove that Novopharm’s drug contained any of the patented polymorph – in this case form I had not disappeared. Novopharm was able to proceed with its generic.
The ‘gastrointestinal infringement’ argument
Disappearing polymorphs played a similar legal role in a second case between SmithKline Beecham (also now part of GSK) and another Canadian generics company Apotex. The drug in question was the antidepressant paroxetine hydrochloride (Paxil), first developed by Danish company Ferrosan as an anhydrous crystal and licenced to SmithKline, which subsequently discovered and patented a new more stable hemihydrous crystalline form, with one bound water molecule for every two drug molecules.
In 1998 Apotex sought approval to market its own drug based on the first less-stable anhydrate, which was out of patent, but SmithKline objected, using the same disappearing polymorph argument. While not strictly polymorphs, SmithKline claimed that the same kind of environmental seeding process would lead to conversion to at least traces of the still in-patent hemihydrate in Apotex’s drug, infringing the patent.
SmithKline even argued that that Apotex would additionally be infringing the patent as its anhydrate tablets were likely to convert to the hemihydrate in a patient’s stomach due to the increased humidity and higher pressure favouring that form – known as ‘gastrointestinal infringement’. While the court did not accept this argument, SmithKline did overall convince the US court of its disappearing polymorph theory in the 2001–3 case.
In the case, experts on both sides gave conflicting reports on whether the phenomenon was occurring in paroxetine, or whether paroxetine anhydrate and hemihydrate could in fact co-exist without one converting into the other. The court again accepted the concept of the disappearing polymorph and sided with SmithKline.
But in a case of ‘be careful what you wish for’, the case took an interesting twist at appeal two years later. Based on SmithKline’s own disappearing polymorph argument, the court found that because the more stable hemihydrate would inevitably be formed when following the method to make the original anhydrous crystal, it was effectively already anticipated by the first patent and the new patent was therefore now invalid
According to Terrence Threlfall, a now retired chemist and expert in polymorphs who acted as expert witness for Apotex, both sides ultimately benefited. ‘Glaxo [then SmithKline] sold £14 billion and kept generics except Apotex off its back. During this time Apotex sold $5 billion so both sides were pleased.’
Polymorph problems beyond pharmaceuticals
These legal issues around disappearing polymorphs were focused on the pharmaceutical industry, but this is not the only sector where they have had an impact. Sun says it has been an issue for his field of inorganic battery materials. One example is lithium iron phosphate, LiFePO₄, which has an alpha and beta phase; alpha is the basis of lithium iron phosphate batteries, used in electric cars and grid storage because its structure is stable during lithium insertion and removal. But it can unexpectedly transform to the beta-phase due to variations in synthesis conditions – acid concentration, reaction time or temperature – which is far less amenable to reversible lithium ion transport, resulting in worse battery performance. ‘This kind of thing happens all the time,’ says Sun.
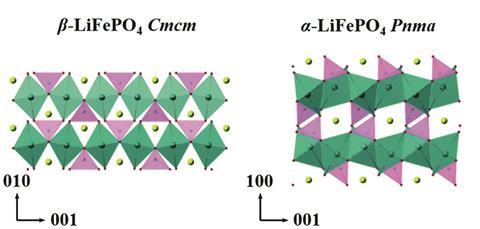
Although unpredictable, so far the alpha polymorph has not become entirely inaccessible and researchers have developed methods to get it back. But Davey says this isn’t that different to the other cases in the pharamceutical industry. ‘I was always of the view that they weren’t really disappearing. It’s just that somehow something had changed. Because there are so many potential variables that might influence what you see in a crystallisation.’
In 2024 Cruz-Cabeza and collaborators published a new method for converting between polymorphs by grinding down crystals in a ball mill. Their study used ritonavir’s two polymorphs as an example. Back in the late 1990s Abbott laboratories had found the faster nucleating metastable form I disappearing, with their synthesis leading only to the more stable but difficult to nucleate form II. But Cruz-Cabeza found after 15 minutes of ball milling form I with a microlitre amount of isopropanol (a technique known as liquid-assisted grinding) she could produce form II. Then milling in water for several hours, form II could be returned to the disappearing form I.
The key to the conversion is to grind the crystals back to the nanoscale, where surface properties start to dominate stability – just as they do in the nucleation phase of crystallisation.‘By going to conditions that give you a steady state of smaller crystal sizes, we were able to go back to form I,’ explains Cruz-Cabeza. She says the result was a surprise because she didn’t think the method would work with a large, complicated molecule like ritonavir. ‘[The molecule] has lots of rotational bonds, which change when going from one polymorph to another so not only do you need to change the way the molecules interact with each other, but you also need to change the conformation significantly.’ But the introduction of mechanochemical energy during milling allows this to happen.
Cruz-Cabeza says this could be ‘eye opening’ for the pharmaceutical industry and how they turn an active compound into a stable solid with the best properties. ‘This simple 15 minute experiment with very simple instruments can really help you discover many polymorphs that might be very relevant for the development of your drug.’
Predicting the unpredictable
Screening polymorphs is a critical part of their process, says der Horst. ‘There is a broad screen of solid states of the chemical compounds, and then they select one … and of course, then hope in the development process that this is not going to disappear.’ He thinks there are probably more cases of disappearing polymorphs than we are aware as companies don’t always want to talk about their failures.
In the past 20 years, better computational modelling is allowing chemists to at least understand what crystal structures are likely to form, but ‘the problem lies in predicting the crystallisation kinetics’, explains der Horst. ‘If you have the crystal structure of two polymorphs, you cannot really say which one crystallises faster.’ This means pharma companies don’t always know if their crystal structure will turns out to be a metastable phase that will one day suddenly disappear in favour of a more stable polymorph.
One way to tell is measuring the nucleation rate of different polymorphs, often simply by counting crystals forming from a super-saturated solution. The nucleation sites themselves are too small to be visually detected. But Sun has been building a computational tool that he hopes will be able to anticipate the nucleation rate of polymorphs and predict when a more stable phase might be likely to arise, as well as help engineer the right conditions to either prolong or shorten the life of a desired metastable form.
He does this by calculating the surface energies of different polymorphs using density functional theory which models the molecules electronic structure. It can be ‘a little tricky’, he says, because surfaces behave differently to bulk materials but he thinks they are close to finding a solution. ’I really believe that in the next year we’ll have made significant strides towards the question of scientifically understanding why disappearing polymorphs happen.’
Rachel Brazil is a science writer based in London, UK
Article updated 22 May 2026 to correct the nationality of Ferrosan

















No comments yet