The materials required in battery-powered cars are providing new challenges to chemists and the chemical industry. Clare Sansom reports
The internal combustion engine is not dead, but it may be beginning to die. One of the few bold steps taken at the November 2021 Cop26 climate conference in Glasgow, UK, was a declaration on phasing out sales of petrol and diesel cars by 2040 in all markets and by 2035 in leading ones: many European countries have set earlier dates, with the UK opting for 2030. Even this timetable seems frustratingly slow to many, but it is probably the earliest that the automotive industry, with its long lead times, can support.
This shift away from petrol and diesel, and from oil and gas more generally, is not only driven by regulation, or even by the climate emergency alone. Cop26 in November 2021 was sandwiched between the UK’s petrol crisis in September 2021 and Russia’s catastrophic invasion of Ukraine. The petrol crisis may have been short-lived and relatively trivial, but few in the UK last autumn will easily forget seeing petrol station after petrol station closed or queuing for hours to fill their fuel tanks. The Ukraine war has brought changes that are less immediate but far more serious and wide-ranging, with our reliance on oil and gas from Russia in particular becoming as much a security as an environmental issue. Furthermore, Russia’s is not the only unpalatable regime to be sitting on large proportions of the world’s remaining fossil fuel reserves; the governments of Saudi Arabia and Qatar, in particular, are hardly more savoury.
For many people a car or van is the only viable solution to their transport needs
These global trends are already accelerating the interest in making the switch away from petrol; about 12–15% of all cars sold in Europe this year are likely to be electric. The Connected Places Catapult is one of nine innovation centres in the UK that work with business on late-stage research and development in a wide range of sectors. Its focus on cities, transport and places links it to the challenge adopted in 2019 of achieving net zero greenhouse emissions by 2050. It is investing heavily in projects to reduce the 27% of the UK’s total emissions that are caused by transport, with an emphasis on low- and zero-emissions vehicles.
‘There is clearly a role for promoting behaviour change, for making it easier for people to choose to walk, cycle or take public transport, and for innovative “mobility as a service” solutions such as the now widely available e-scooters,’ says Alan Nettleton, a senior technologist at Connected Places Catapult. ‘But for many people a car or van is the only viable solution to their transport needs. It is therefore essential that we make sure that private vehicles are as clean as possible, and that means battery electric vehicles.’
Whether the cars of the future are autonomous or manually driven, and whether they are individually owned or shared, they will almost inevitably be electric. And while, obviously enough, fully electric vehicles consume no hydrocarbon fuel, their manufacture requires many more fine chemicals, plastics and polymers than conventional vehicles.
Demanding criteria
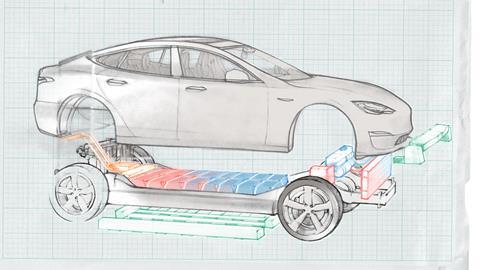
Battery electric vehicles (often called BEVs) have a powerful electric traction motor to replace the internal combustion engine, and no fuel pump, fuel line or fuel tank. It therefore has no exhaust or tailpipe, and so no ‘tailpipe emissions’ (a key statistic used to rate the emissions or otherwise of other vehicle types). This contrasts with various forms of hybrid electric cars, which although containing a battery, still have petrol or diesel engines. A large battery pack made up of numerous lithium-ion cells and positioned under the floor of the car drives the motor, and power from the motor is transferred to the wheels via an electric transmission. A coolant system surrounds the electrical and electronic components of the vehicle to insulate them and maintain a safe operating temperature.
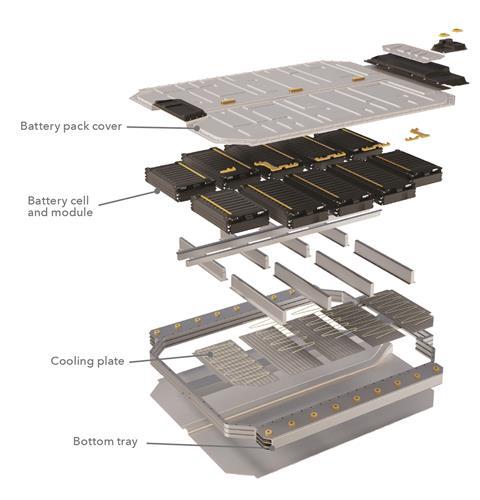
An electric vehicle can only perform safely and efficiently if the materials used in its construction have a particular set of properties, both alone and in combination. Parts of the vehicle are subject to extremely high voltages, temperatures and stresses, so some components must be completely electrically isolated, and the vehicle must also be resistant to such stresses. The battery must be protected from crash damage to prevent fires, and the whole vehicle must be made from materials that are low in density to reduce the total vehicle weight. This problem limited the take-up of the earliest models. The materials must also be as durable and inexpensive as possible. ‘So far, BEVs have only penetrated the high end of the market,’ says Nettleton. ‘Manufacturing costs must come down, but universal take-up is unlikely to be possible until the vehicles are commonly available second-hand.’
And finally, even something as seemingly mundane as colour can be a problem: the pigments used to indicate common hazards such as high voltage cables must be stable. It is therefore not surprising that some of the world’s top chemical companies rapidly moved into this sector.
Fluoropolymers are ideal materials for electrically insulative coatings
The lithium-ion cells in today’s vehicles pack much more punch than the ubiquitous batteries of the same type found in our handheld electronic devices. ‘The packaging of the battery in the floor of the car, and the separators between the cells, must offer complete insulation and protection not only during normal use but also during the catastrophic failure that might occur after a crash,’ says Paul Shearing, a professor of chemical engineering at University College London and a member of the SafeBatt consortium, a collaboration of six UK universities and industrial partners funded by the Faraday Institution.
Furthermore, the trend is for higher power density in the components of the motor and drivetrain. This requires motors with improved winding wire properties, smaller motors with the same power and lower cost through design simplicity and the use of less materials to enable more torque and speed. The current standard wire coatings are easily degraded by heat, high voltage and motor frequency with loss of their insulating and electrical properties. New classes of fluoropolymers are seen as the next generation of materials which have ideal properties for these applications.
The Japanese multinational chemicals company Daikin Industries originally specialised in air conditioning, meaning it is well placed to enter this market because of its expertise in fluorocarbon chemistry. ‘Fluoropolymers are ideal materials for electrically insulative coatings for motor winding applications because of the unique nature of the carbon–fluorine bond,’ says Peter Hupfield, vice-president for application development at Daikin Chemical Europe Innovation Center in Dortmund, Germany. ‘This is one of the strongest covalent bonds in chemistry because of its polar nature, with high electron density around the fluorine atom, so it is very difficult to break. These compounds are exceptionally stable and as such provide unique electrical, mechanical, chemical and thermal resistance properties, ideal for such demanding applications as enabling the development of higher performance wire.’
Safety concerns
The German-based chemicals giant BASF is something of an anomaly as both a specialist in the automotive sector and a pioneer in sustainability: for example, in 2008 it was the first large manufacturing company in the world to record and publish its own carbon dioxide emissions. The materials chemistry of electric vehicles is a perfect fit for this company, and BASF products are used in the bodies of electric vehicles and also as coolants, anti-corrosion coatings, and – perhaps surprisingly – specialist colourants.

All cars need coolants to prevent their engines from overheating and potentially sparking fires, but those in electric cars are particularly important because of the higher temperatures and voltages involved. A typical coolant used in the automotive industry consists largely of a mixture of water and ethylene glycol. Both liquids are needed because of their characteristic physical properties: water because its high heat capacity and thermal conductivity allows the rapid dissipation of heat from the battery throughout the vehicle, and glycol because of its low melting point, which is crucial if the vehicle is to be used in a cold climate. A 1:1 mixture of water and glycol will not freeze until the temperature reaches –37°C. Other components of coolants, at little more than trace levels, are organic or inorganic corrosion inhibitors added to protect the metallic components of the coolant circuits – aluminium, copper, steel or other alloys – from damage.
BASF is developing a new, safer generation of coolants with lower electrical conductivity to protect against the possibility of electrical shocks and fires. ‘Our new Glysantin Electrified coolants will not react with the high-voltage components of the battery even if that battery is damaged in an accident and the coolant comes into direct contact with it’, says Itamar Malkowsky, senior manager for technical marketing automotive fluids at BASF in Germany. ‘We achieve this by using glycol derivatives with lower polarity and either non-ionic corrosion inhibitors, or ionic ones with a low ion dissociation tendency.’
Since the 1940s, when the RAL colour chart system was completed, any of 40 standardised colours can be specified using the letters RAL followed by a four-digit code. ‘A few years ago, a working group of the German automotive industry approached us to ask for a bright and well-recognised warning colour to characterise high-voltage components of electric vehicles’, says Josef Wünsch, senior vice-president of R&D for performance materials at BASF. ‘They asked for RAL2003, an easily recognisable tint of orange [described in the colour list as Pastel orange]; our challenge was to produce a formulation that would remain stable throughout the life of the vehicle.’ Shades of orange produced from simple mixtures of red and yellow pigments can rapidly fade to brownish, but BASF’s researchers have been able to produce a mixture of inorganic pigments and organic dyes that matches the specification exactly and stands the test of time.
Knauf Industries, founded in 1932 in Germany and now operating throughout Europe, is another chemicals company that has found a role in electric vehicle manufacture. Knauf’s lightweight, durable expanded polypropylene and expanded polystyrene foams have been used to make moulded vehicle components for over 20 years. These materials, however, also have physical properties that make them ideal as cell separators within a battery pack. They are excellent thermal and electrical insulators, which greatly reduces the risk of heat escape or current flow between battery parts or outside them even if the car is involved in a collision.
Recycling requirements
Vehicles with no tailpipe emissions can be assumed to be net zero during their lifetimes, at least if they are powered by renewable energy (which is not under the control of either the manufacturer or, necessarily, the driver). They can only be considered completely net zero, however, if no emissions arise from their manufacture and disposal, and if their components are recycled. The batteries, at least, have proved to last much longer than industry experts originally expected. ‘A typical lithium-ion battery will remain “efficient enough” through up to 1000 charging cycles’, says Shearing. ‘With a typical range of about 250 miles between charges, this comes out at a quarter of a million miles… few conventional cars do this mileage, so the battery may last longer than the car.’ In fact, a battery is ‘retired’ at about 80% of its original capacity, when it still has many potential uses, just not in cars. The batteries that eventually replace lithium-ion ones should be even more efficient and long-lasting.
With a life of 1000 charging cycles, the battery may last longer than the car
Large commercial vehicles represent a particular challenge to manufacturers, as their powerful batteries need more resources to keep them charged. A typical lorry will need a 500kWh battery, which is about eight times the capacity of one that drives a typical medium-sized private car. ‘For efficient freight delivery, batteries in electric lorries must be able to run without recharging for as long as their drivers are legally allowed to drive before a rest break, which is 4.5 hours’, says Nettleton. ‘They must also be recharged quickly, which puts further pressure on the charging stations.’ However, alternative solutions for climate-friendly road freight are now being considered. Siemens’ eHighway, which is being trialled in Germany on a few stretches of autobahn, provides electricity to trucks through an overhead pantograph similar to those used by trains.
Cars and lorries will be part of our lives for decades to come. If we are to truly meet the challenges of net zero (and beyond) in time to avert the worst climate breakdowns, and if we are to wean ourselves off imports of fossil fuels from unstable or autocratic regimes, the next generation of electric cars must be more sustainable ‘from cradle to grave’, with as many components as possible easily recycled. And perhaps above all, they must be made more affordable for the average motorist. One important step forward will be the replacement of the lithium-ion battery technology with one or more battery types that use materials that are cheap, recyclable and readily available. Meeting the technical requirements for these and other materials to be used in future electric vehicles is bound to provide even more opportunities for chemists.
Clare Sansom is a science writer based in Cambridge, UK











No comments yet