An osmium sandwich complex with two boron-based rings is the first true example of a carbon-free ferrocene analogue. The compound was synthesised by researchers in India who say that this complex improves our understanding of metal-ligand bonding.
The classic ferrocene sandwich complex consists of an iron atom coordinated between two cyclopentadienyl rings. In 2002, chemists isolated a titanium species coordinated to two five-membered phosphorus rings, though this only partially resembled ferrocene due to its anionic character.
Researchers have now created a true carbon-free analogue of ferrocene by reacting a square planar osmium complex with borane dimethylsulfide. The neutral complex features a central osmium atom sandwiched between two five-membered boron rings (B5H10).
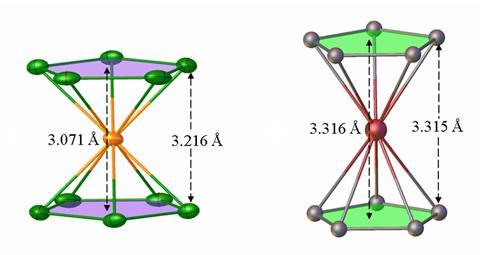
X-ray crystallography revealed that the rings are slightly rotated relative to one another, compared to ferrocene’s eclipsed geometry. The distance between the rings in the osmium analogue is also shorter than in ferrocene. The researchers say that is due to the larger boron ring interacting with the osmium’s more diffuse orbitals, as well as bridging hydrogens that force the rings closer together.
The team also isolated another osmium complex in which one boron-based ring connected to the central osmium atom was perpendicular to the other ring, as well as its ruthenium analogue. The researchers say that comparing these complexes ‘brings greater understanding of metal-ligand bonding and this has implications in intercalations in graphene and borophenes’.
References
S Mohapatra et al, Science, 2026, DOI: 10.1126/science.aed9192















No comments yet