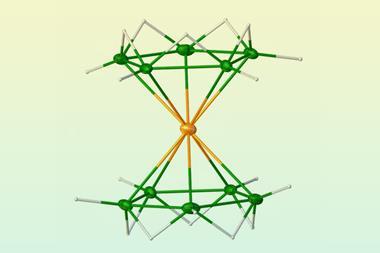
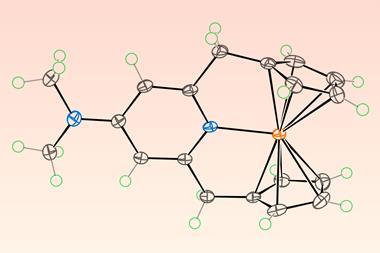
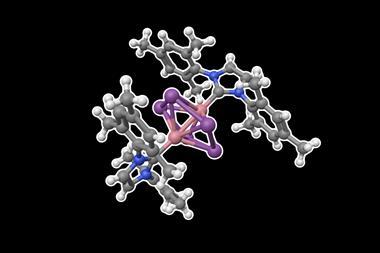
Stable sandwich complexes in which metal atoms are coordinated between two cyclopentadienyl ligands have now been made with all of the metals in the top row of the periodic table’s d-block, after US researchers isolated a copper metallocene. The compound comprises a copper atom sandwiched between two ligands that feature bulky substituents.
Ferrocene, an iron atom coordinated between two cyclopentadienyl ligands, was famously discovered in the 1950s and led to a surge in interest in sandwich complexes. Soon after ferrocene’s structure was deduced, analogues featuring most of the other 3d transition metals were isolated. A sandwich complex of titanium was finally isolated in 1998, while those featuring scandium were reported for the first time two years ago – leaving copper as the only 3d metal yet to be trapped between cyclopentadienyl ligands as a stable metallocene.
Now, researchers at the University of California, Irvine and Davis have finally done so. They used a derivative of the cyclopentadienyl ligand featuring three bulky tert-butyl substituents. The team suggests that attractive interactions between the bulky substituents on opposite ligands help to stabilise the complex and notes that other research groups have used the ligand previously in sandwich complexes featuring a variety of 3d metals, rare-earth metals and actinides.
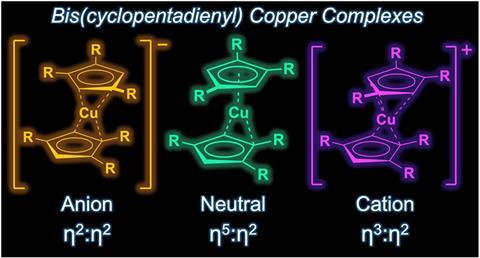
While two anionic sandwich complexes featuring copper were reported in 1995, these were extremely unstable. This was likely due to the tendency of copper to promote carbon–carbon bond forming reactions, something that affects all sandwich complexes involving this metal.
The newly reported copper metallocene forms blue–green crystals that are stable under ambient conditions. The researchers also produced a colourless anionic version of the complex, as well as a purple cationic species.
References
J D Queen et al, J. Am. Chem. Soc., DOI: 10.1021/jacs.5c19366











No comments yet