New bond describes interactions between group 11 nanoparticles and Lewis bases
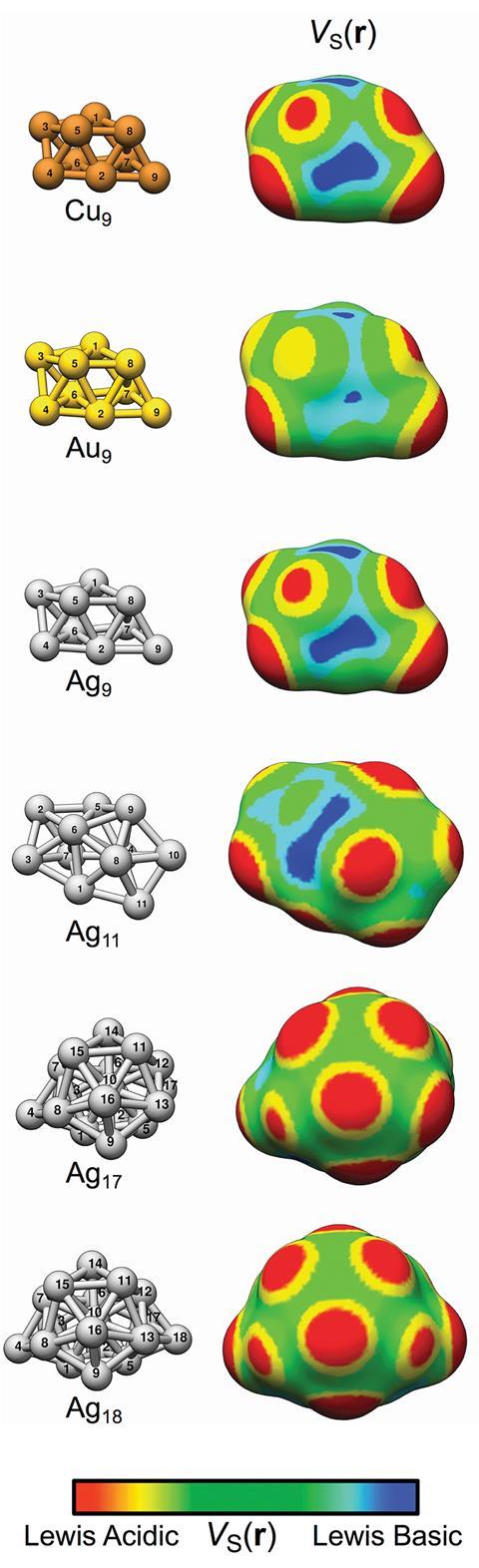
Researchers in Sweden have described a new type of chemical bond and proposed that chemists call interactions between copper, silver and gold nanoparticle and electron donors ‘regium bonds’. This concept could aid scientists developing new heterogeneous catalysts.
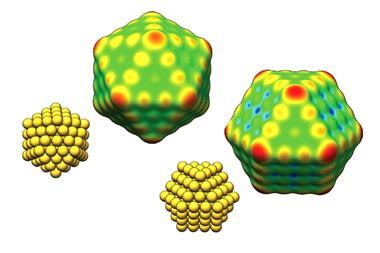
The work continues the team’s computational research on nanostructural effects in catalysis. In the previous work, they looked at gold nanoparticles. Now, they have extended the study to copper and silver clusters. ‘We found that the unique catalytic properties of nanoparticles of gold, silver and copper clusters are due to regions of positive surface electrostatic potential at sites that bind Lewis bases,’ says Tore Brinck of KTH Royal Institute of Technology in Stockholm, who led the work.
σ-hole theory is well established and describes regions of low electron density that have a positive surface electrostatic potential and can attract electron-rich atoms such as Lewis bases. This same theory rationalises why halogens bind Lewis bases. In this study, Brinck and co-workers identified σ-lumps, which are regions of enhanced electron density with negative surface electrostatic potential that arise because of σ-holes. σ-lumps on a nanoparticle’s surface become binding sites for Lewis acids. ‘This is not strange. If you take away electron density from one part of the cluster or molecule it must go somewhere and generate a region of increased electron density,’ explains Brinck.
‘We have decided to introduce the term regium bonding since gold, silver and copper interact with Lewis bases in a similar manner, and the similarity of the interactions is a consequence of the metal atoms having the same type of valence electron configutaion, ie a half-filled s-orbital,’ says Brinck. Regium bonding is similar to halogen and hydrogen bonding, where Lewis bases or hydrogen atoms also have half-filled s-orbitals.
‘Chemists tend to think that bonding and interactions of metals and metal nanoclusters are very different from those of normal molecules. It is important to see the similarities, so that we can use knowledge from traditional chemistry in the design of a better catalyst,’ says Brinck. Understanding how bonding depends on the electronic structure of clusters is important for heterogeneous catalysis. ‘If we can understand and predict this bonding, we can design more efficient catalysts,’ he adds.
‘This discovery is important, as a deep understanding of the physical nature of regium bonds will facilitate the design and synthesis of new nanoparticles with tunable properties,’ says σ-hole expert Antonio Frontera from the University of the Balearic Islands in Spain. ‘Then, the surface of these new nanoparticles can be adjusted to catalyse a variety of chemical reactions.’
Brinck and co-workers plan to investigate other nanoparticle clusters to see how chemical environments affect the strength of regium bonds.
References
This article is open access
J H Stenlid, A J Johansson and T Brinck, Phys. Chem. Chem. Phys., 2018, DOI: 10.1039/c7cp06259a







No comments yet