This paper was retracted on 2 September 2024.
Researchers in the US and China have synthesised a new carbon allotrope – γ-graphyne – in bulk for the first time. The new way to prepare this elusive material offers scientists the opportunity to fully investigate its unique electronic and mechanical properties.
Carbon exists in many different forms. The most well-known allotropes are naturally occurring diamond and graphite, while the laboratory synthesis of fullerenes and graphene were both rewarded with Nobel prizes. Carbon nanotubes (CNTs) are yet another form whose interesting physical properties sparked a whole field of research. But all of these allotropes are based on single types of carbon atom – graphite, graphene, fullerenes and CNTs all feature sp2-hybridised carbon atoms where each atom is bonded to three others, while diamond is a network of sp3-hybridised atoms with each carbon forming single bonds to four adjacent atoms.
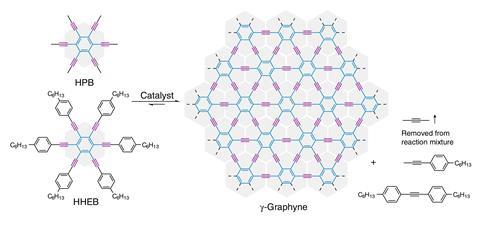
But there are many other possible forms of carbon that involve combinations of different types of carbon atom. γ-graphyne is one such example and includes both sp2 and sp-hybridised carbons – it is essentially a network of aromatic rings separated from each other by pairs of triple-bonded carbon atoms. While γ-graphyne was first predicted to be stable several decades ago, it has previously only been synthesised in small fragments many of which lack long-range order.
Now, a team led by Yingjie Zhao from Qingdao University of Science and Technology and Wei Zhang from the University of Colorado Boulder has developed a new way to synthesise γ-graphyne based on a reversible alkyne metathesis reaction using alkylnyl-substituted benzene monomers. The method produced crystalline flakes of the material containing around 30 layers of γ-graphyne stacked together. The largest flake had an area over 10μm2.










No comments yet