A simple new theory that can explain why a metal forms a particular structure has been developed.1 The method allows researchers to understand and predict structures in solid compounds and alloys over a wide range of conditions. ‘This theory is based on our finding that the electrons in many metals occupy so-called quasi-atom orbitals, which are local quantum orbitals centred at the voids between atoms,’ explains Russell Hemley at the University of Illinois Chicago in the US, who led the study together with Maosheng Miao from California State University, Northridge. ‘The chemical interactions between such localised electrons control the metal structures,’ he adds.
‘This new chemical perspective of metals is [an] alternative to the traditional free electron gas model provided by physicists,’ comments Álvaro Lobato, a theoretical chemist at the Complutense University of Madrid in Spain who wasn’t involved in the study. He says that in the new chemical model, the metals behave like inorganic crystals with positive cores and quasi atoms interacting as cations and anions.
‘Determining the metal structures and their pattern across the periodic table is a puzzle that has remained unsolved since the first structure of metals was detected by x-ray diffraction about 100 years ago,’ points out Hemley. He notes that the structures of several classes of metals still can’t be described by the ‘physics’ or band–structure model. ‘Even though we can explain many advanced properties of metals and other compounds, such as superconductivity, we don’t understand why some metals crystallise in the face-centred cubic structure, others take on the hexagonal close-packed structure and some crystallise as body-centred cubic.’
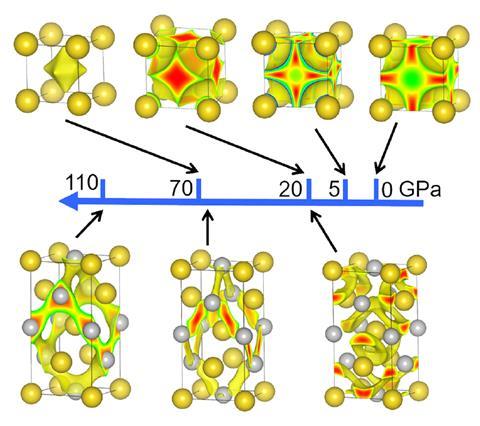
But the structural changes that happen when metals are put under pressure are even more puzzling, adds Hemley. ‘Many metals give up high-symmetry structures and transform to lower symmetry and large voids, so there are many high-pressure structures that aren’t close-packed, which is counter-intuitive.’
‘These structural transformations are caused by an effect that we call sublattice interactions,’ says Miao. He explains that the metal lattice can be split into two sublattices. ‘If the electron localisations of the sublattices match, the intercalation of the sublattices stabilises the structure, but if the electron localisations cause repulsive interactions between the two sublattices, that will destabilise the whole structure,’ he says. ‘Under high enough pressure, all high-symmetry structures become repulsive, so the metal has no choice but to adopt structures with larger voids and lower symmetry.’
To understand different systems, the team carried out precise quantum mechanics calculations that included well-defined electron states and performed simulations of many metals’ structures across the periodic table, including under compression. They carefully examined the electronic states of many metal lattices with varying metals, lattices and sizes and analysed the electron localisation.
José Manuel Recio, a materials scientist at the University of Oviedo in Spain, says that the new theory represents a fundamental advance in the understanding of materials at the atomic level because of its general perspective. ‘Not only for crystalline solids under ambient conditions, but also for its extension to extreme pressure and temperature conditions.’
The researchers were originally looking for a simple chemical explanation for the existence of high-pressure electrides, a surprising phenomenon in which some alkali metals can become transparent insulators under pressure. ‘Investigating this question led us to a simple theory that works for many other elemental metals and compounds under ambient conditions,’ says Hemley. ‘This is an example of how the study of matter under extreme conditions can inform us about chemistry and materials under normal or more familiar conditions.’
Miao points out that the theory has some limitations as metal structures are the result of many factors, including temperature, the nuclear quantum effect and magnetic interactions. ‘None of these are included in the current approach,’ he adds. ‘However, in most of these more complex cases, the local electron chemistry is still the major contribution to structural preferences.’
The new approach, which can be used to predict the behaviours of different types of solids including superhydrides, low-dimensional materials, intermetallics and ionic compounds, could help scientists to find materials with exciting properties. ‘Our conceptual framework and the related theory for compounds can be used to search for and predict new metal superhydrides that might superconduct under high temperatures and lower pressure,’ says Miao.
References
Y Sun et al, Proc. Natl. Acad. Sci. USA, 2022, DOI: 10.1073/pnas.2218405120











No comments yet