Water’s almost unique feature of expanding on freezing can cause water droplets to break apart, and new research from the US has provided important insights into the factors that affect this and the resulting ice.1 The findings could potentially lead to advances in atmospheric modelling.
Water does not instantly solidify as soon as its temperature drops below 0°C because ice crystals must first nucleate and spread. Water droplets can therefore remain in a metastable supercooled liquid that is stable well below freezing. When water does eventually start to freeze, it is most likely to do so near the outside of a droplet. This is partly because simple geometry dictates that a 3D shape has more area farther from its centre, and also because a cooling droplet cools on the outside first. If the outside freezes first, the centre then freezing creates pressure in the ice.
This pressure can be released if the solid shell cracks, allowing liquid water to escape from the interior. This then freezes into protuberances called spicules. The droplet can also shatter completely, leaving behind multiple chunks of ice. In 2017, Sander Wildeman and colleagues at the University of Twente in the Netherlands observed using high speed imaging that droplets larger than around 50μm were likely to shatter, whereas smaller droplets were not.2
In the new work, however, Claudiu Stan of Rutgers University in New Jersey and colleagues looked at more deeply supercooled droplets just 40μm in size. The researchers used Stanford’s Linear Accelerator Laboratory to perform simultaneous x-ray diffraction imaging and optical microscopy on thousands of droplets between 6.4 and 7.8ms after injection into a vacuum chamber. They found that around 60% of them exploded. The researchers suspect this may be because, in a more deeply supercooled environment, the droplets form a stronger shell of ice, preventing water escaping to form spicules: ‘Our explanation at present is that maybe we are keeping and building the pressure because of the increased strength of the shell,’ says Stan. He hopes that putting the work together with that of others might allow the field to arrive at a formula connecting air pressure, temperature and droplet diameter with the probability of a droplet splitting and the number of fragments produced. This is a long-standing problem in cloud physics, for example.
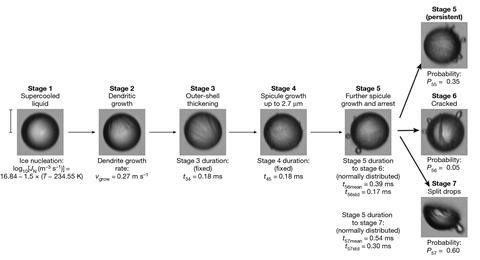
Analysing the x-ray diffraction patterns, the researchers found that the water did not freeze into familiar hexagonal ice. A previous experiment involving supercooled water droplets from an oil–water emulsion observed that, minutes after freezing, the ice was in an intermediate phase, with stacking defects connecting regions of hexagonal ice and regions of cubic ice.3 However, in the new experiment, the frozen droplets adopted a strained hexagonal configuration 1ms after freezing. ‘There is no continuous experiment between these two and there are many orders of magnitude difference in timescale,’ says Stan. ‘It seems that the strain would be sufficient to introduce at least some stacking defects, but the mechanism through which this would happen we do not know.’ The researchers are now planning to extend their work to longer timescales to try to observe the evolution of the structure.
Computational chemist Pablo Debenedetti of Princeton University in the US is impressed by the researchers’ combination of experimental techniques and says their work is ‘an interesting approach to the very complex approach of droplet dynamics under rapid freezing conditions.’ He now hopes they will push towards even colder temperatures, where, he says, ‘I would be interested in whether they see signatures of cubic ice.’ He also suggests they might apply the techniques to other liquids.
References
1 A Kalita et al, Nature, 2023, DOI: 10.1038/s41586-023-06283-2
2 S Wildeman et al, Phys. Rev. Lett., 2017, 118, 084101 (DOI: 10.1103/PhysRevLett.118.084101)
3 B J Murray and A K Bertram, Phys. Chem. Chem. Phys., 2006, 8, 186 (DOI: 10.1039/b513480c)















No comments yet