India faces urgent demand for advanced wound care technologies
This essay focuses on the policy gaps in regulating advanced wound care dressings in India, particularly those involving tissue-engineered and multifunctional materials. It highlights the absence of harmonised quality control parameters across global regulators, fragmented clinical trial standards, and inadequate integration of IP protections with innovation pathways. With India importing most of such products and lacking a clear framework to support indigenous innovation, the essay proposes an anticipatory policy model. This includes adaptive clinical trial protocols, IP incentives for bio-material innovation, and affordability mechanisms to enable local production, with strategic relevance for both civilian healthcare and military applications.
Advanced wound care technologies, including hydrogel, hydrocolloid, foam, tissue regeneration-based dressings and negative-pressure wound therapy (NPWT), mark a significant chemistry-driven trend in wound management. Unlike traditional dressings, these modalities actively regulate moisture balance, reduce infection risks and support tissue regeneration. Globally, innovations in biomaterials and nanotechnology are pushing the field toward bioactive and sensor-integrated ‘smart dressings,’ which represent the next frontier of personalised care.1–3
In India, the demand for such solutions is urgent. With one of the world’s largest diabetic populations, a rapidly ageing society and widening disparities in healthcare access, the incidence of chronic wounds such as diabetic foot ulcers and pressure injuries is rising sharply. However, the development and adoption of advanced wound care products remain limited. Key barriers include the absence of product-specific regulatory guidance, fragmented intellectual property (IP) frameworks that discourage innovation and limited clinical research infrastructure to generate local evidence.4,5 These gaps result in slow translation of laboratory discoveries into clinical practice, continued dependence on costly imports and restricted access for patients in low-resource settings.6
The core policy gap, therefore, lies in the misalignment between India’s clinical needs and its regulatory and innovation ecosystems. While advanced wound care technologies promise transformative benefits, India lacks foresight-driven mechanisms to regulate, scale and sustain them. This paper advances the concept of ‘policy foresight’, a framework that anticipates emerging requirements in wound care while ensuring regulatory robustness, affordability and innovation incentives. The key questions addressed are:
- How can India design regulatory pathways that balance innovation with patient safety?
- What mechanisms can strengthen IP protection while enabling equitable access?
- How can local clinical research capacity be expanded to generate robust, affordable evidence?
By addressing these, India can foster domestic innovation, reduce import dependence and improve equity in advanced wound care delivery.
Context and rationale
Market trends and burden of disease
Advanced wound care products, including hydrogels, hydrocolloids, collagen dressings and NPWT are increasingly important in India for managing both chronic wounds (diabetic foot ulcers, venous leg ulcers, pressure injuries) and acute injuries from burns, trauma and surgery. With over 77 million people living with diabetes, the lifetime risk of developing foot ulcers ranges from 15–25%, representing a substantial clinical and economic challenge. Additional contributors, such as high rates of road accidents, surgical site infections and immobility-related ulcers, add to the burden.7,8
The market reflects this demand. Valued at INR22.16 billion (£180 million) in 2024, it is projected to grow to INR30.79 billion by 2030, growing by 6.4% a year. Growth drivers include wider healthcare access, rising awareness of modern wound care and penetration into tier II and tier III cities. Increasing adoption of NPWT, demand for portable home-care devices and interest in affordable, locally manufactured dressings highlight both opportunities and unmet needs.
Technological landscape and policy gap
Emerging technologies
Globally, wound dressings have advanced from passive coverings to multifunctional therapeutic platforms. Developments in material science, nanotechnology and regenerative medicine have enabled dressings with sensing, antimicrobial and bioactive delivery functions.
Sensor-integrated dressings now employ flexible electronic platforms to continuously monitor wound parameters such as pH, moisture and temperature, enabling early infection detection and guiding personalised therapeutic decisions.9
Bioactive hydrogels are being engineered as delivery reservoirs for growth factors, exosomes and nanoparticle constructs; preclinical evaluations demonstrate their capacity to accelerate angiogenesis and re-epithelialisation.
Gas-modulating systems, including topical nitric oxide and oxygen-delivery devices, are showing promise in enhancing vascularisation and disrupting biofilms, thereby addressing two key barriers to chronic wound healing.10
Antibiotic-free antimicrobial strategies, such as stimuli-responsive hydrogels and reactive oxygen species (ROS)-sensitive nanofibers, offer localised infection control while mitigating the risk of antimicrobial resistance. Living skin substitutes, exemplified by products like Apligraf (approved in the US), integrate cellular components with biomaterial scaffolds to promote tissue regeneration, illustrating the frontier of personalised wound care.9 These advances position advanced dressings not as adjuncts but as integral components of regenerative and precision medicine.7
Regulatory gaps
Despite similarities with EU and US frameworks, India’s regulatory system for wound dressings lacks clarity in several areas, the same has been described in the table below.
Table 1: Gap matrix: regulation of advanced wound care technologies
| Dimension | India (MDR 2017) | EU (MDR 2017/745) | US (FDA) | Gap in India |
|---|---|---|---|---|
|
Device classification |
Classes A–D based on wound depth; no explicit provisions for bioactive, absorbable or sensor‑integrated dressings |
Rule 21 up‑classifies substance‑based dressings; stricter risk categorisation |
Risk‑based Class I–III; bioactive/sensor‑based dressings reviewed under higher classes or via De Novo |
Ambiguity for novel dressings creates regulatory uncertainty for innovators |
|
Clinical evidence |
Pilot and pivotal trials required, but no product‑specific guidance for NPWT, bioactive hydrogels or living substitutes; foreign data waivers common |
Mandatory clinical evaluation and post‑market clinical follow‑up (PMCF) |
Clinical trials or bridging studies required for novel features; strong guidance on wound‑care devices |
Weak local evidence base and over‑reliance on foreign data |
|
Registries and infrastructure |
No national wound registries; limited accredited clinical investigation sites; weak systematic data capture |
National and international wound registries in use; stronger clinical research networks |
Specialised registries (e.g. wound care, diabetic foot ulcers) and multi‑site clinical research infrastructure |
Lack of registries and accredited sites limits real‑world evidence and local trial capacity |
|
Post‑market oversight |
Materiovigilance Programme of India (MvPI) present but underutilised; no binding PMCF requirement |
PMCF mandatory; periodic safety update reports (PSURs) required |
Post‑market surveillance, recalls and mandatory reporting enforced |
Weak post‑market surveillance and absence of PMCF reduce ability to detect safety and performance issues |
|
Quality standards |
Partial alignment with EN ISO 14155:2020; enforcement inconsistent |
Fully aligned with ISO 14155:2020 GCP for medical device trials |
FDA mandates GCP‑compliant trials; strong enforcement mechanisms |
Trial quality and regulatory credibility compromised by partial standards alignment |
Without lifecycle evidence requirements, India risks approving products with inadequate safety and performance data, discouraging both innovation and clinician confidence.11
Intellectual property barriers
Robust IP protection is crucial for fostering domestic innovation, yet Indian innovators face hurdles. Section 3(d) of the Patents Act restricts claims on incremental innovations, limiting the scope for polymer gels, coatings or antimicrobial additives. Other exclusions (3(c), 3(i), 3(j)) constrain biologic dressings or microbiome-based approaches. Patent prosecution is often slow, costly, and dependent on specialised expertise that many SMEs lack. Unlike the US and EU, where biologic and combination dressings can obtain clearer protection, India’s framework leaves innovators uncertain about freedom to operate. Weak IP awareness, high filing costs and limited technology transfer mechanisms further restrict commercialisation. Without complementary regulatory clarity, innovators hesitate to commit to long-term IP strategies, perpetuating dependence on imports.12
Strategic importance and national needs
Defence and emergency medicine
For India’s military, advanced wound dressings are strategically important. In battlefield conditions, dressings with haemostatic, antimicrobial and nanotechnology-enabled properties can reduce mortality and accelerate recovery. Domestic products such as Axiostat and Hemohalt illustrate indigenous capability, yet broader adoption requires cost-effectiveness, biocompatibility and ease of application.
Import dependency and innovation gaps
Currently, a significant proportion of high-value dressings are imported, delaying access and inflating costs. Regulatory and manufacturing challenges limit local capacity to produce complex bioactive or sensor-based dressings. Experiences from other sectors suggest that reducing import dependence will require sustained policy support, investment in R&D infrastructure and harmonised quality standards.
Risks, opportunities and urgency
Risks: Continued ambiguity may discourage domestic startups, prolong import dependence and restrict access for low-resource populations.
Opportunities: India can foster innovation by introducing product-specific regulatory guidance, aligning IP and clinical frameworks, and building dedicated wound research networks.
Urgency: With rising diabetes and vascular disease, chronic wounds are becoming a public health challenge. Anticipatory governance through foresight-driven regulation, IP reform, and investment in evidence generation is essential to ensure equitable access and national self-reliance.
The policy idea
Policy proposal: centre for advanced wound care policy and innovation
This proposal introduces the Centre for Advanced Wound Care Policy and Innovation (CAWCPI), a permanent, multi-stakeholder centre under the Ministry of Health and Family Welfare, designed to provide regulatory foresight, foster innovation, strengthen IP strategy and ensure affordable access to advanced wound care technologies. CAWCPI would function as a centre of excellence in policy foresight, bridging regulators, industry, academia, and defence healthcare to reshape India’s wound care landscape.
Policy concept
CAWCPI is poised to serve as India’s national hub for anticipatory governance in advanced wound care. Its mission will be threefold:
- Develop product-specific regulatory pathways tailored to advanced wound technologies.
- Design harmonised testing and standards frameworks that provide clarity for innovators.
- Integrate IP support, affordability safeguards, and public–private innovation hubs into the regulatory ecosystem.
- Promote advanced research in wound care devices
By unifying these functions, CAWCPI will shift India from a reactive regulatory stance to a proactive, foresight-driven model that anticipates emerging challenges before they reach the market.
Institutional structure
CAWCPI would be semi-autonomous, functioning with its own scientific advisory board, regulatory experts and innovation fellows. This ensures independence while remaining closely aligned with CDSCO and BIS mandates .
Functions of CAWCPI
-
Regulatory foresight and guidance
- Draft product-specific regulatory guidelines for hydrogels, NPWT devices, bioactive scaffolds and sensor-based dressings
- Define proportionate clinical evidence requirements, bench testing for simple dressings, bridging studies for sensor-based products, and full trials for regenerative substitutes
- Publish advanced guidance notes to give innovators clarity before filing applications
-
Standards and testing frameworks
- Develop Indian standards for biocompatibility, antimicrobial efficacy, exudate absorption and digital sensor accuracy
- Harmonise with ISO 10993 and EN MDR standards to ensure international acceptability
- Create accredited testing hubs across India to reduce reliance on overseas labs
-
IP and innovation support
- Provide patent landscaping and advisory services to startups and SMEs
- Support innovators in navigating Section 3(d) and related exclusions of the Patents Act
- Establish model licensing frameworks that encourage affordability without discouraging commercialisation
-
Clinical evidence ecosystem
- Coordinate multicentric trials aligned with ISO 14155 GCP
- Reduce reliance on foreign data waivers by creating local evidence roadmaps
- Mandate post-market clinical follow-up (PMCF) studies, like EU requirements, but adapted for Indian conditions
-
Public–private innovation hubs
- Build regional innovation hubs linking universities, hospitals, contract manufacturers and SMEs
- Offer shared infrastructure for preclinical testing and prototyping
- Enable co-funded translational research projects targeting low-cost wound care solutions
- Create more contract manufacturing facilities to support small-scale industries
Distinctiveness of the policy
India’s current system under IMDR (2017) provides only broad classifications (Class A–D) and permits data waivers for imported products. There is no specialised framework for advanced wound technologies. By contrast, CAWCPI introduces a permanent foresight-driven institution, not just a regulatory office tasked with shaping the future of wound care in India.
Globally, EU MDR mandates clinical evaluation and PMCF, and the US FDA applies device-specific ‘special controls’. However, neither system integrates affordability safeguards or IP advisory into its regulatory models. CAWCPI would be unique in explicitly combining regulatory clarity, IP protection, affordability strategies and domestic manufacturing promotion within one institutional platform.
Anticipatory governance role
CAWCPI’s foresight function ensures that regulatory planning is not limited to current technologies but anticipates future trends and challenges. Its activities would include:
- Horizon scanning to systematically identify emerging innovations such as gas-modulating hydrogels, stimuli-responsive dressings, and AI-integrated wound sensors.
- Scenario planning workshops with regulators, clinicians, industry and patient groups to anticipate regulatory, ethical and affordability dilemmas before technologies reach the market.
- Strategic procurement alignment so that products critical for battlefield medicine and public health emergencies are prioritised for accelerated review and adoption.
- Global benchmarking to align Indian policies with evolving EU, US and IMDRF frameworks, ensuring both international credibility and domestic adaptability.
This proactive stance would prevent regulatory lag, reduce litigation and position India as a global leader in wound care governance.
Expected Impact
The establishment of CAWCPI is expected to deliver wide-ranging benefits across multiple levels. For innovators, it will provide clearer approval timelines, lower compliance costs, access to IP advisory and patent landscaping support, and shared infrastructure through regional innovation hubs. For the healthcare system, the centre will promote affordable, domestically produced wound dressings, reduce reliance on costly imports and enhance clinical trial capacity and evidence quality. Patients will benefit from wider access to modern therapies, particularly in tier II and III cities, along with reduced risks of amputations and complications from chronic wounds, and greater confidence in product safety through lifecycle monitoring. At a strategic level, CAWCPI will ensure a reliable supply of haemostatic and antimicrobial dressings for defence healthcare, strengthen resilience for trauma management in disaster situations and advance long-term national self-reliance in critical biomedical technologies.
Implementation Strategy
Phased Introduction
The CAWCPI should be established through a three-phase approach to ensure smooth integration into India’s regulatory and innovation ecosystem.
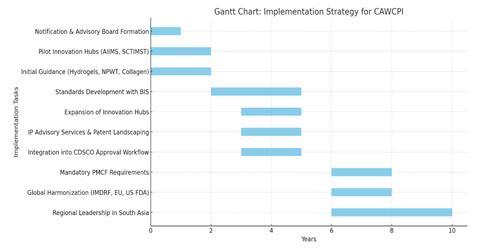
Phase I: Establishment and pilot (year 1–2)
- Notification of CAWCPI under the Ministry of Health & Family Welfare as a semi-autonomous body linked to CDSCO.
- Formation of a scientific advisory board with experts in biomaterials, clinical wound care, IP law and regulatory affairs.
- Development of initial product-specific regulatory guidance for three priority categories: hydrogels, NPWT systems tissue-engineered-based dressings.
- Launch of two pilot innovation hubs in collaboration with premier institutes (eg, AIIMS, SCTIMST) to test shared research and evaluation infrastructure.
Phase II: Expansion and standardisation (year 3–5)
- Rollout of harmonised Indian standards for wound dressings in partnership with BIS.
- Expansion of innovation hubs to four additional regions, ensuring coverage in both metro and tier II/III cities.
- Integration of CAWCPI’s guidance into CDSCO approval workflows, reducing ambiguity for startups and manufacturers.
- Introduction of IP advisory services and patent landscaping tools for SMEs.
Phase III: Consolidation and global alignment (year 6 onward)
- Mandatory post-market clinical follow-up (PMCF) and lifecycle evidence requirements for all advanced wound products.
- Strategic partnerships with IMDRF, EU and US FDA for global harmonisation.
- Positioning CAWCPI as a regional centre of excellence in South Asia, supporting neighbouring countries with regulatory and innovation frameworks.
Key stakeholders
- Regulators: Ministry of Health & Family Welfare, CDSCO, BIS.
- Research bodies: ICMR, DBT, academic medical centres and national institutes of biomedical technology.
- Industry and startups: Domestic manufacturers, SMEs developing wound care products, multinational firms seeking local approvals.
- Civil society and patients: Advocacy groups, diabetes associations, rural health NGOs ensuring patient perspectives and access equity.
- Strategic sectors: Ministry of Defence and disaster response agencies for battlefield and emergency applications.
- CAWCPI’s advisory board should formalise representation from each stakeholder group, ensuring decisions reflect a balance of innovation, safety, and affordability.
Anticipated barriers and mitigation
Institutional inertia: Resistance to a new body within existing structures may slow uptake.
Solution: Embed CAWCPI as a semi-autonomous centre of excellence with statutory backing under CDSCO, while ensuring alignment with IMDR. Regular joint reviews can ease integration.
Data gaps: India lacks high-quality local clinical evidence on wound care outcomes.
Solution: Mandate structured clinical registries for diabetic ulcers, burns, and trauma cases, linked with innovation hubs. Incentivise hospitals through funding support to participate in PMCF studies.
Capacity constraints: Few specialised inspectors and trial investigators have experience with advanced biomaterials or sensor-based dressings.
Solution: Develop training modules on ISO 14155 GCP, wound-specific device testing and materiovigilance. Establish fellowships for young regulators and clinicians to build expertise.
IP and commercialisation hurdles: SMEs often struggle with patent filing and enforcement.
Solution: Create a wound care IP facilitation cell within CAWCPI to support drafting, filing, and technology transfer, reducing costs for innovators.
Affordability concerns: Advanced wound dressings may remain inaccessible to low-resource patients.
Solution: Introduce tiered pricing models and link procurement of domestically manufactured products with government reimbursement schemes like Ayushman Bharat. Promote more cost-effectiveness studies.13
Enabling mechanisms
Legal framework
CAWCPI can be notified under the Medical Device Rules (MDR, 2017) through an amendment, granting it authority to issue regulatory guidance and set mandatory testing standards. Statutory recognition ensures its decisions are binding and enforceable.
Timelines
Incentives
- Tax credits or grant-in-aid schemes for startups conducting local trials.
- Priority review status for innovators following CAWCPI guidance.
- Public procurement preference for products tested and validated under CAWCPI standards.
Capacity building
- Annual training programs for regulators, clinicians and industry scientists.
- Specialised training programs for caregivers and hospital staff on advanced wound care products and their usage
- Development of e-learning modules in collaboration with BIS and WHO.
Monitoring and evaluation
- Establish a performance dashboard tracking approval timelines, number of patents supported, local manufacturing growth and patient access outcomes.
- Independent audits every three years to assess CAWCPI’s effectiveness.
Risks and ethics
The establishment of the CAWCPI introduces new opportunities, but also carries ethical dilemmas, social risks and potential unintended consequences that must be acknowledged early.
One significant ethical challenge is equity of access. Advanced wound care technologies are often expensive, and without deliberate safeguards, CAWCPI could unintentionally accelerate innovation that benefits urban, affluent populations while excluding patients in tier II and III cities or rural regions. This creates a risk of deepening health inequities. To mitigate this, affordability criteria and tiered pricing mechanisms must be integrated into policy design, ensuring public procurement prioritises locally manufactured, cost-effective products.
Another ethical concern relates to data governance. With increased emphasis on clinical trials, registries and sensor-based wound care systems, issues of patient privacy, informed consent and responsible data sharing become paramount. Misuse of sensitive health data could erode public trust. Embedding transparent consent processes, strict data anonymisation protocols and third-party audits into CAWCPI’s clinical evidence frameworks will safeguard against this.
Intellectual property dilemmas also arise. While stronger IP protection encourages innovation, aggressive exclusivity could stifle competition and inflate prices. CAWCPI must therefore balance patent support with mechanisms for compulsory licensing and open innovation in areas of public health priority.14
Institutional risks include bureaucratic inertia and the possibility that CAWCPI could become another layer of regulation rather than a facilitator of innovation. This could discourage startups or create duplicate approval steps. Safeguards against this include lean governance, public dashboards tracking approval timelines and independent external evaluations.
Social resistance may also emerge if clinicians and patients perceive advanced wound products as ‘elite technologies’ or if traditional methods are sidelined without adequate evidence. Embedding clinician training, patient education campaigns and participatory feedback forums will normalise the adoption of new dressings.
To embed trust, equity, and transparency, CAWCPI must operate with clear communication channels: publishing regulatory guidance notes openly, engaging civil society groups in advisory boards and institutionalising stakeholder consultations. In doing so, it will not only manage risks but also build legitimacy as a trusted, patient-centred regulatory body.
Conclusion
The establishment of the CAWCPI offers India an opportunity to transform a fragmented and import-dependent sector into a model of foresight-driven governance. By integrating regulatory clarity, product-specific standards, IP support and public–private innovation hubs, CAWCPI will address long-standing barriers that have constrained the growth of advanced wound care in the country. Its design ensures that innovation is not only encouraged but also aligned with affordability and accessibility, directly benefiting patients across diverse healthcare settings.
From a national perspective, CAWCPI will serve as a catalyst for clean growth and self-reliance, reducing dependence on costly imports while stimulating indigenous manufacturing and research. By embedding foresight into policy – through horizon scanning, scenario planning and global benchmarking – India can position itself as a regional leader in anticipatory biomedical governance, bridging gaps between science, policy and public health. The focus on lifecycle evidence, equitable access and strategic defence applications further underscores its holistic relevance.
In reaffirming India’s capacity for science leadership, CAWCPI demonstrates how proactive institutional design can balance innovation with public good. It embodies a governance capability that not only meets today’s wound care challenges but also anticipates tomorrow’s, ensuring resilience in the face of emerging biomedical and demographic pressures. With CAWCPI, India can chart a path that is innovative, inclusive, and globally respected, setting a precedent for policy foresight in advanced healthcare technologies.
References
1 A Alberts et al, Gels, 2025, 11, 123 (DOI: 10.3390/gels11020123)
2 H E Gültekin et al, Mater Horiz, 2024, 11, 363 (DOI: 10.1039/D3MH01330H)
3 A Alberts et al, Gels, 2025, 11, 271 (DOI: 10.3390/gels11040271)
4 V Yadav et al, J. Wound Care, 2019, 28, S32 (DOI: 10.12968/jowc.2019.28.Sup8.S32)
5 H Vieira et al, J. Mar. Pol., 2025, 173, 106575 (DOI: 10.1016/j.marpol.2024.106575)
6 B Sheokand et al, J. Polym. Sci., 2023, 61, 1389 (DOI: 10.1002/pol.20220734)
7 P Mallanagoudra et al, Polymers, 2025, 17, 2303 (DOI: 10.3390/polym17172303)
8 A S Sidhu and V Harbuzova, Front. Clin. Diabetes Healthc., 2024, 5, 1440209 (DOI: 10.3389/fcdhc.2024.1440209)
9 F Mariani et al, ACS Sens., 2021, 6, 2366 (DOI: 10.1021/acssensors.1c00552)
10 X Han et al, Nat. Commun., 2024, 15, 3435 (DOI: 10.1038/s41467-024-47696-5)
11 K R Harkin, J Sorensen & S Thomas, Int. J. Technol. Assess. Health Care, 2024 , 5, e2 (DOI: 10.1017/S026646232300274X)
12 J Bergsland, O J Elle and E Fosse, Med. Devices: Evidence Res., 2014, 7, 205 (DOI: 10.2147/ MDER.S43369)
13 T Yaniv, D Beeckman and A Gefen, J. Tissue Viability, 2024, 33, 938 (DOI: 10.1016/j.jtv.2024.10.001)
14 S Igras et al, Global Public Health, 2021, 16, 882 (DOI: 10.1080/17441692.2020.1820550)

















No comments yet