
Sunlight can be used to convert polystyrene waste and excess sulfur into valuable organic products. The team that created the new process hopes that it could be a way to tackle the millions of tonnes of polystyrene waste generated every year, without the need for energy intensive methods.
The chemical industry produces over 20 million tonnes of polystyrene annually for food packaging, electronics and insulation. Yet less than 1% of polystyrene is recycled each year. Various methods – such as electrolysis, oxidation and pyrolysis – offer ways to recycle this plastic, but these often require large amounts of energy or are inefficient, says study leader Qing-An Chen at the Dalian Institute of Chemical Physics in China.
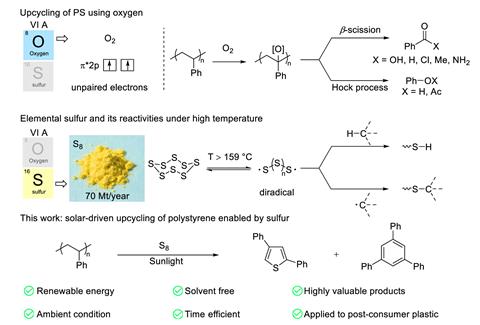
‘By combining elemental sulfur in excess supply with non-degradable polystyrene waste, and driving the transformation via clean solar energy, we address both plastic pollution and the efficient utilisation of sulfur resources,’ explains Chen. Elemental sulfur is a byproduct of crude oil refining, with several million tonnes of the element piling up each year.

Exposing polystyrene and elemental sulfur to sunlight for as little as two minutes under ambient conditions generated several organic products. This included a diphenylated thiophene, used in semiconductor materials, and 1,3,5-triphenylbenzene, a versatile organic building block that can cost up to $400 (£300) per kg.
This method was able to upcycle various post-consumer waste products, including spoons, cups, food packing, falcon tubes and assay plates.
Mechanistic studies revealed that energy from sunlight generates sulfur radicals that are then able to abstract hydrogen atoms from the polystyrene backbone. Hydrated sulfur radicals then react with the polymer product through a series of steps to create the final compounds.
‘The selectivity and yield are some of the biggest challenges for this work,’ says Andrew Dove, a polymer chemist at the University of Birmingham in the UK. ‘Separation is usually energy intensive or not done very sustainably… so getting [the selectivity] better to be able to avoid separation would be an important target.’
A large proportion of the final reaction mixture is partially degraded polystyrene – up to around 40% by weight – though the team found this product could be used to fully depolymerise polystyrene or act as a UV-blocking additive in polystyrene films.
Chen explains that the team is now working on scaling up the upcycling process, as well as better understanding the mechanism behind the conversion. ‘We [also] aim to extend this method to other common plastics, such as polyethylene, polypropylene and polyvinyl chloride,’ he adds.
References
Y Liu et al, J. Am. Chem. Soc., 2026, DOI: 10.1021/jacs.6c01318
















No comments yet