The controversial process of enhanced enzyme diffusion could enable living systems to resist local equilibrium

Over the past decade or so, the notion of enzymes as biological catalysts has got complicated. Many experiments have produced evidence that something odd can happen to enzymes, ranging from urease to glycolytic enzymes to polymerases, as they catalyse their intended reactions: the protein molecules seem to diffuse more rapidly than they do while inactive.1–4
To say that this ‘enhanced enzyme diffusion’ (EED) has provoked a lot of argument is something of an understatement. Several explanations for it have been proposed,5 ranging from electrophoresis1 to conformational changes6 and convection.7 And yet the case for EED is still disputed in the first place.8 For one thing, some of the experiments involve averaging over many molecules, while for others the interpretation is complicated by the enzymes being multimeric, so it’s not clear if one is tracking movement of the whole enzyme or just a part.
Still, if we suppose EED is real, how is it even possible? It seems that the enzyme is somehow tapping into the free energy released in the reaction it catalyses,9 rather than that energy being simply dissipated. In other words, the enzyme seems to be doing what Maxwell’s demon does. That conclusion has now been demonstrated theoretically by Kunihiko Kaneko, an expert in complex systems at the University of Copenhagen, Denmark, and his colleagues.10
What’s the problem with Maxwell’s demon?
Maxwell’s demon – the impish being imagined by James Clerk Maxwell in 1867 – could undermine the second law of thermodynamics by engineering a non-equilibrium system from one at equilibrium, thereby creating a reservoir of energy that could be tapped to do work. Equivalently, the demon made entropy decrease. In Maxwell’s scenario, the demon operates a trapdoor separating two gas-filled compartments, letting molecules through selectively so that all those with high kinetic energy (‘hot’) accumulate on one side and those with low kinetic energy (‘cold’) gather on the other. So starting from a gas at uniform temperature, the demon creates a temperature gradient that can be harnessed for work. It’s essentially a perpetual-motion device.
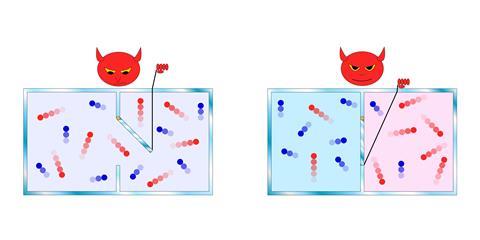
It took a century to figure out the flaw in this apparent violation of thermodynamics. The demon has to store information about the molecules in its memory – but it’s a finite memory, and so it needs to be erased from time to time to keep the process going. But it turns out that erasure has an entropic cost, repaying the entropic deficit accumulated while the demon does its work.11 In this way, Maxwell’s demon revealed a deep link between thermodynamics and information: specifically, information itself can be converted to heat, acting as a kind of fuel.
Life’s equilibrium
So the demon can only defy the second law temporarily. All the same, some biochemists in the early 20th century, such as Frederick Donnan and Archibald Hill12 wondered if living systems might act like Maxwell’s demon in staving off equilibrium (meaning death) for a while. Oddly, Erwin Schrödinger made no mention of this possibility when he pondered how organisms seem to defy the second law in his 1944 book What Is Life?, even though he corresponded extensively with Donnan since the mid-1930s.
Kaneko and colleagues have shown how enzymes can act as thermodynamic ‘information engines’ of the same kind. The key criterion for EED in their scheme is that it occurs (transiently) only when the enzyme catalyses the forward reaction, and not the reverse reaction. You could say that it exploits this asymmetry somewhat like a ratchet, in a manner analogous to the stochastic ratcheting of some motor proteins along an asymmetric potential,13 another biochemical process compared with Maxwell’s demon. The enzyme’s enhanced diffusion acts as a kind of memory of its having catalysed the forward reaction. By using this information, the enzyme can extract work from the system. Under some conditions it can displace the steady-state concentrations of the reactant and product away from chemical equilibrium. (Crucially, there is an energy source driving the process, and so there is energy dissipation overall: the second law is not really broken.)
Kaneko says that the process suggests a way that living systems could use the reaction energy to locally resist equilibrium. The enzyme still catalyses the reaction, but in a way that keeps nudging it away from its equilibrium state. Might this help to address Schrödinger’s conundrum? That’s far from clear, but it’s an intriguing thought.
References
1. H S Muddana et al, J. Am. Chem. Soc., 2010, 132, 2110 (DOI: 10.1021/ja908773a)
2. S Sengupta et al, ACS Nano , 2014, 8, 2410 (DOI: 10.1021/nn405963x)
3. C Riedel et al, Nature, 2015, 517, 227 (DOI: 10.1038/nature14043)
4. A Y Jee et al, Proc. Natl. Acad. Sci. USA, 2017, 115, 141 (DOI: 10.1073/pnas.1717844115)
5. R Golestanian, Phys. Rev. Lett., 2015, 115, 108102 (DOI: 10.1103/PhysRevLett.115.108102)
6. X Bai and PG Wolynes, J. Chem. Phys., 2015, 143, 165101 (DOI: 10.1063/1.4933424)
7. T S C MacDonald et al, Angew. Chem., 2019, 58, 18864 (DOI: 10.1002/anie.201910968)
8. M Feng and MK Gilson, Annu. Rev. Biophys., 2020, 49, 87 (DOI: 10.1146/annurev-biophys-121219-081535)
9. A-Y Jee, T Tlusty and S Granick, Proc. Natl. Acad. Sci. USA, 2020, 117, 29435 (DOI: 10.1073/pnas.2019810117)
10. S Ichii, TS Hatakeyama and K Kaneko, Phys. Rev. Lett., 2026, 136, 038401 (DOI: 10.1103/flv6-zw1v)
11. R Landauer, Phys. World, 1999, 12, 37 (DOI: 10.1088/2058-7058/12/1/26)
12. J Needham, Q. Rev. Biol., 1928, 3, 77
13. R D Astumian, Commun. Phys., 2025, 8, 365 (DOI: 10.1038/s42005-025-02303-x)
















No comments yet