How to scavenge platinum group metals from laboratory waste
Precious metals are one of the big workhorses of chemistry. Cross couplings, metathesis and photochemistry all depend on these rare and expensive catalysts, not to mention the wider commercial demand for industrial synthesis, automotive catalytic converters and green energy technologies. The environmental footprint of extracting and disposing of these materials is huge, but rarely given much thought by typical lab scientists. ‘Most of the time, our goal is to make sure that you get the purest and most high-yielding reaction,’ says catalysis researcher Shiao Chow from the University of Strathclyde, UK. ‘You know it’s a precious metal but there’s not a strong awareness of the need to recover and you don’t really consider that it should be part of the protocol when it comes to synthesis.’
A selective scavenger

For new academic Chow, it was partnership with Johnson Matthey through their Platinum Group Metals Award Scheme which sparked the inspiration to challenge this status quo. The company provided the group with 10g of platinum group metals to investigate biocompatible catalysis, requesting that any metal waste be returned for recycling in house. ‘It’s an amazing initiative but I realised there was a gap in the chemistry – I can perform biocompatible chemistry with the catalyst, Johnson Matthey will pick up the waste matter from me, but I didn’t have a method to recover the metals from my reactions,’ Chow explains.
With funding from the Royal Society of Chemistry’s Sustainable Laboratories grant, she set about developing a method to recover spent metal directly from the reaction mixture. (Intellectual property concerns mean Chow cannot currently reveal the identity of the platinum group metal species.) After carefully designing selectively chelating ligands, she grafted these motifs onto a solid support, creating an easily-separated metal scavenger which could then be sent to Johnson Matthey for reformulation into fresh reagent.
But Chow quickly realised that the recovered platinum group metal also had potential as a catalyst in its own right and the scavenged metal, still affixed to the solid support, effectively mediated several further reactions before the material was ultimately recycled. These early results are extremely exciting, but the scavenger is still at a prototype stage, says Chow. She is currently investigating the performance of the system on more complex waste mixtures supplied by departmental colleagues and will use this feedback to optimise the scavenger, and any future analogues, to make this a user-friendly method suitable for anyone working in catalysis.
‘This project has made me more conscious of how we perform chemistry in a lab and it’s really made me think about how I can transform my workflow,’ Chow concludes. ‘The dream is that this reusable catalyst design will be useful in industry where they have a strong interest in solid phase catalysis. We are now working on attracting engagement and investment from interested parties.’
Car parts to catalysts

In contrast, finding ways to valorise used precious metals has always been the subject of research in James Wilton-Ely’s group at Imperial College London, UK. His team, alongside longstanding collaborator Angela Serpe at the University of Cagliari, Italy, works on the recovery of palladium from catalytic converters, transforming these old car parts into useful catalytic reagents.
‘Angela has come up with a very nice method of using iodine and tetrabutylammonium iodide to treat catalytic converter waste and make it into a palladium dimer,’ he says. ‘We can then use this complex as a starting point for catalysts which can do all these fundamental palladium-catalysed C–C, C–N, and C–O bond-forming reactions.’
This recovery chemistry is well-established and already at the pilot plant stage. But, for Wilton-Ely, this raised the question of whether the same method could be translated to laboratory palladium waste. Unlike the well-defined composition of a catalytic converter, pots of reaction leftovers and old reagents are something of an unknown quantity and the team was initially unsure how the optimised conditions would tolerate these messy mixtures.
To begin, the researchers therefore focused exclusively on expired reagents, collecting roughly 40g of oxidised palladium tetrakis from around the department and subjecting it to Serpe’s recovery conditions. ‘We found that we could make the palladium dimer, or that we could make other palladium complexes that were also catalytically useful,’ Wilton-Ely explains. ‘That was a nice starting point as we immediately had an approach which could use what was otherwise going to be thrown away.’
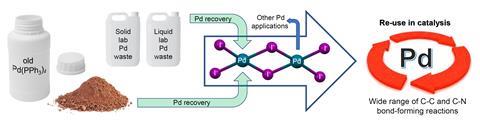
Emboldened by this success, they repeated this process on a model waste stream. Researchers were asked to make a note of the approximate composition of waste at the point of disposal, enabling the team to estimate the rough quantities of reagents needed for recycling. ‘This process recovered the palladium dimer in around 50% notional yield. It might not seem that high, but it’s 50% that would otherwise have just gone to waste,’ he says.
The method is both mild and safe, with the dimer conveniently precipitating out of the mixture for easy separation. The biggest challenge, says Wilton-Ely, is popularising the slightly unusual palladium dimer catalyst. ‘It’s a dimer, which is already a bit unusual, and it’s got these iodide ligands where people would tend to expect a chloride,’ he explains. ‘Our catalyst looks a bit different in structure but we’ve shown that it behaves in the same way, and in many ways, it’s actually superior to these traditional catalysts.’
Tips to start recovering metal waste
- Start small. Look at the local issues in your own lab and how you could transform an aspect of your workflow to make it more environmentally friendly.
- Establish protocols within your lab for how to store and record different types of metallic waste.
- Be creative – there’s no need to return whatever you’re recovering back into exactly the same material.
- Engage with others, both within your university and from external organisations, as they may be able to offer you funding, materials, or support.

How chemists are making laboratories more sustainable

A collection of articles sharing tips from researchers who reduced their environmental impact with support from the RSC’s Sustainable Laboratories grants
- 1
- 2
- 3
- 4
- 5
 Currently
reading
Currently
reading
Sustainable laboratories: Recovering metals
- 7





























No comments yet