From ancient folk remedy to the wonder drug of the early industrial age and beyond. Mike Sutton traces the remarkable history of aspirin

Around 250 years ago, a riverside walk was the first step in the story of asprin. The story takes in the rise of the global pharmaceutical industry, two world wars, one Nobel prize and the drug is still the most widely taken in the world, with new therapeutic uses still emerging. But it all started when Edward Stone announced that powdered willow bark was a helpful treatment for malaria.
This pharmaceutical revolution had a long prehistory. Ancient Sumerian and Egyptian texts recommended willow bark for various complaints, and Greek, Roman and Islamic medical authors noted its power to relieve pain and reduce fevers. However, its tendency to cause inflammation – and occasionally bleeding – of the stomach lining considerably diminished its utility. Possibly for this reason, medieval and early modern European physicians usually ignored it (though in some areas it survived as a folk remedy).
Once the botanical resources of the Americas were accessible, Peruvian quinine became the preferred treatment for fevers. But quinine was expensive, so Stone was pleased to find a locally available remedy for the malarial fever – or ‘ague’, as it was then called – which afflicted him. His success encouraged other sufferers to try it, even though he was merely an amateur scientist without medical qualifications.
Stone had joined the Anglican clergy after graduating from the University of Oxford, eventually becoming chaplain to Sir Jonathan Cope at Chipping Norton in Oxfordshire. During the 1754 parliamentary election, Stone acted as the agent for a local candidate whose father was the Earl of Macclesfield – a noted astronomer, and president of the Royal Society. It was Stone’s letter to the Earl that communicated his discovery to the Society. Published in the 1763 Transactions, it reveals an interesting blend of scientific and pre-scientific reasoning.
Ague, Stone noted, was generally associated with marshy environments and their bad air (mal aria) – this was long before mosquitoes were identified as the vectors of malaria. He was also aware of the ancient herbalists’ doctrine of signatures: ‘many natural maladies carry their cures along with them, or that their remedies lie not far from their causes’. And so, during a riverside walk in 1757, he tried chewing some willow bark. Since its bitter taste resembled that of quinine, he hoped it might relieve his discomfort.
Stone then decided to pursue a more systematic approach. After drying the bark over a baker’s oven, he pounded and sieved it thoroughly. Then, by taking gradually increasing doses of it, he showed that two scruples (about 2.5g) every four hours dispelled his fever. Over the next five years he treated about fifty other sufferers with his preparation, and although it could not eradicate their malaria entirely, it gave welcome relief from the symptoms.
From tree to pharmacy
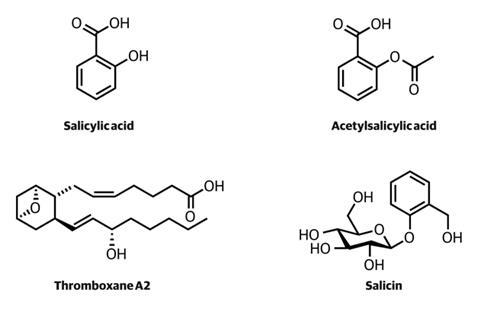
After Stone’s results were published, some apothecaries began using willow bark to treat fevers, and in 1828 the German chemist Joseph Buchner isolated its active ingredient. He called the yellowish, bitter-tasting substance salicin (salix being the Latin for willow).
Two years later Johann Pagenstecher, a Swiss apothecary, extracted a useful pain-reliever from another folk remedy: the meadowsweet plant whose botanical name, spiraea, later suggested the brand name aspirin. Soon afterwards, the German chemist Karl Löwig found that its active component was identical to Buchner’s salicin, and in 1838 Raffaele Pirea, an Italian working in the Paris laboratory of Jean-Baptiste Dumas, succeeded in converting salicin to salicylic acid.
This compound (2-hydroxybenzenecarboxylic acid) proved to have similar therapeutic properties to salicin, but unfortunately it also irritated the stomachs of some patients. In 1853, the French chemist Charles Gerhardt prepared its acetyl ester by treating sodium salicylate with acetyl chloride. This compound eventually proved to be a far more useful remedy, but Gerhardt’s reaction was difficult to manage and a practicable synthesis remained elusive for many years.
Meanwhile, salicin from plants was still prescribed by innovative doctors like Thomas Maclagan, who treated rheumatic fever successfully with it in the 1870s. However, once the mass production of synthetic pharmaceutical products began, acetyl salicylic acid (ASA) played a key role in the industry’s expansion, largely thanks to the efforts of the chemical company Bayer.
The emergence of Bayer
Friedrich Bayer was one of several German chemical manufacturers inspired by William Perkin’s discovery of synthetic aniline dyes. In 1863, a century after Stone’s publication, Bayer and his partner Johann Weskott established a new dyestuff company which was moderately profitable (though seldom innovative) until his death in 1880. Things changed in 1884 when Carl Rumpff – Bayer’s son-in-law and successor as head of the firm – hired an ambitious young chemist named Carl Duisberg.
After completing his doctorate in chemistry at Jena, Duisberg had been sidelined by a year of compulsory military service and he was eager to resume laboratory work. His achievements at the bench were so impressive that he quickly became Bayer’s head of research. When Rumpff died in 1890, Duisberg was effectively in control of the company, and he soon began transforming it.
Bayer’s business skills would have accomplished little without an effective product.
By then, Bayer had already followed other German chemical manufacturers into the pharmaceutical business. The early synthetic drugs were tested haphazardly, and often failed to fulfil all the marketing claims made on their behalf. But in 1899 – largely thanks to Duisberg’s more systematic approach – Bayer hit the jackpot with a sophisticated version of Stone’s homely medicine.

Duisberg had organised the company’s research workers into specialised divisions. The pharmaceutical group (supervised by Arthur Eichengrün) developed new drugs, while the pharmacology group (led by Heinrich Dreser) tested them. In 1897 Eichengrün was seeking a compound similar to salicin, but without its unpleasant side-effects. His assistant, Felix Hoffmann, succeeded in improving on Gerhardt’s synthesis of ASA by heating salicylic acid with acetic anhydride under reflux, distilling off the unwanted acetic acid, and then crystallising pure ASA from benzene.
Dreser distrusted the product and refused to test it, but Eichengrün sent samples to a colleague in Berlin who had numerous medical contacts. At first Dreser dismissed the positive results of these unofficial trials, but after Duisberg endorsed them he backed down. The new drug – probably at Eichengrün’s suggestion – was marketed as Aspirin, which soon became the everyday name for acetyl salicylic acid in many languages.
How the credit for this achievement should be distributed has been a subject of some debate. However, it is clear that both commercial and medical factors contributed to the drug’s success. Aggressive advertising hammered aspirin’s reassuringly non-technical name into the public consciousness, while astute lawyers defended its trademark status in every important marketplace. Nevertheless, Duisberg’s business skills would have accomplished little without an effective product.
The first wonder drug

Aspirin’s main asset was that it had similar analgesic, anti-inflammatory and fever-reducing powers to salicylic acid, but was less harmful to the stomach. The acetyl ester was not hydrolysed until it reached the alkaline environment of the small intestine, where the resulting acid did less damage. Moreover, unlike traditional opiate pain-relievers – or modern derivatives such as morphine and heroin – ASA was not addictive.
When the first world war ended, aspirin was becoming fully integrated into western culture – handing a tablet to someone in distress seemed as natural as offering them a drink or a cigarette. The influenza pandemic of 1918–19 underlined the drug’s value, but also highlighted its limitations. For many sufferers it bought time while their immune systems manufactured antibodies to defeat the virus. For others, however, it merely prolonged the agony. And medical historians now believe some victims died of internal bleeding after taking excessive doses.
Many of Bayer’s assets were seized as part of the war reparations demanded by the victorious allies. But despite the hyperinflation of the 1920s and the depression of the 1930s, Duisberg doggedly rebuilt the firm. He also played a key role in negotiating the merger of several German chemical companies which created the massive IG Farben conglomerate in 1925.
Like many German businesses in that troubled era, IG Farben contributed to the funds of any political party that had the power do it some good – or harm. But with the rise of the Nazis this voluntary insurance became compulsory blackmail, and after Duisberg’s death in 1935 IG Farben was fully integrated into the Hitler regime. Jewish employees (including Eichengrün) were dismissed, and one of the group’s subsidiaries produced Zyklon B, the poison used in the extermination camps.
At the Nuremburg trials, several of IG Farben’s senior executives were convicted of war crimes, and the conglomerate was broken up. Its pharmaceutical division was then re-established under the old Bayer name, and continued manufacturing aspirin profitably. Meanwhile rival companies prospered with remedies like Disprin and Alka-Seltzer, which blended ASA with weak alkalis to reduce its impact on the digestive system – a particular advantage for hangover victims.
In the 1950s and 60s ASA’s dominant position was challenged by paracetamol and ibuprofen. Neither of them caused the stomach problems associated with salicylates, though both had some negative side-effects of their own. Furthermore, the use of aspirin by children was discouraged in the 1980s because of a suspected link with Reye’s syndrome – a very rare but extremely unpleasant juvenile illness. Yet as aspirin’s popularity was declining, pharmacologists began to understand how it functioned in the body – and then to discover new uses for it.
Sticking around
The groundwork was done by Harry Collier in the Parke-Davis company’s London laboratory. In 1958, he began a series of experiments which demonstrated that aspirin’s analgesic effect was exerted locally, rather than – as previously believed – on the central nervous system. Another received opinion he demolished was that acetylation simply reduced the gastric irritation caused by salicylic acid. Collier showed that ASA was not merely less harmful but also more effective than the acid itself. However, his lab lacked the sophisticated facilities needed to take this work further.

These resources were available at London’s Royal College of Surgeons, where Sir John Vane (assisted by Collier’s former assistant, Priscilla Piper) took on the challenge. Vane and his team had developed ultra-sensitive bioassay methods for tracking the metabolism of substances in a variety of tissues. They were particularly interested in a group of hormones known as prostaglandins – lipid compounds containing twenty carbon atoms (including a five-membered ring).
Their results suggested to Vane that ‘any tissue which was distorted or disturbed or traumatised’ would release prostaglandins. In April 1971 he began to suspect that aspirin’s therapeutic powers might be due to its capacity to inhibit the production of prostaglandins in stressed tissues. An ingenious series of experiments proved this to be the case, and in 1982 Vane shared the Nobel prize in physiology or medicine with Sune Bergström and Bengt Samuelsson, who had also done important work on prostaglandins.
Besides illuminating the biochemistry behind a familiar medicine, this line of research also generated a new therapeutic initiative. ASA was shown to block the production of thromboxane, a lipid which encourages the clotting of blood through its action on platelets.
Clots are essential for healing wounds, but can cause cardiac failure or strokes if they get stuck in the blood vessels of the heart or the brain. In the 1980s extensive trials revealed that regular doses of aspirin significantly reduced the incidence of these circulatory disorders, opening a new market for a drug that had seemed to be on its way out.
Today, scientists are still pursuing fresh therapeutic uses for aspirin – for example, recent research indicates that it may be effective against some varieties of cancer.
And so, two and a half centuries after the riverside rambles of an Oxfordshire clergyman re-acquainted medical science with the therapeutic power of salicylate compounds, aspirin may still have more surprises in store for us.
Mike Sutton is a visiting fellow in the department of humanities at Northumbria University, UK
Further Reading
• Bayer Company History: http://bit.ly/X4Klel
• D Jeffreys Aspirin: The Remarkable Story of a Wonder Drug, Bloomsbury Publishing, New York, 2005
• E Stone, Philos. Trans. R. Soc. 1763, 53, 195 (DOI: 10.1098/rstl.1763.0033)
• Sir John Vane’s Nobel prize lecture: http://bit.ly/T48jXo












No comments yet