Christopher Ingold's insights into mechanism and reactivity established many of the principles of organic chemistry. John Ridd reveals more about his life and work

Sir Christopher Ingold fundamentally changed the approach of chemists to organic chemistry. Many have forgotten what a strange and mysterious subject organic chemistry appeared to be in the days before reaction mechanisms were revealed. Organic chemistry is now viewed in a very different way - based on firmly established mechanistic principles - and, although many people have contributed to that change, almost all chemists would now accept that Ingold was the predominant figure. Although this account focuses on his work in organic chemistry, Ingold also made important contributions to spectroscopy and inorganic chemistry.
Ingold was born in London on 28 October 1893. His father, a silk merchant, was in very poor health with tuberculosis; it was for this reason that the family moved to the supposedly gentler climate of Shanklin in the Isle of Wight. But his father died when Ingold was five years old.
After studying at the local school in Shanklin, Ingold moved to the new secondary school at Sandown where he obtained a county scholarship to study at the new Hartley University College in Southampton. After obtaining his BSc degree there, he moved to Imperial College London and, in 1914, joined the research group of Jocelyn Thorpe, a new professor of organic chemistry. In 1915, he was asked to carry out work in Glasgow on the preparation of gases for use in chemical warfare. This was dangerous work for which Ingold was awarded the British Empire Medal. He subsequently made no reference to the award or to his work during the war.
Award-winning chemistry
In 1920 Ingold returned to Imperial College as a demonstrator with his own research laboratory. It was the beginning of a remarkably active period of research: he published 12 papers in 1921, 16 papers in 1922, and 14 papers in 1923. Most of these dealt with classical organic chemistry, and some with physical chemistry, including specific heats and vapour pressures.
Ingold’s impressive publication record led to rapid movement up the academic ladder - he was promoted to a lectureship, gained a DSc (doctor of science) degree and was awarded the first Meldola Medal in 1922 - an award to commemorate Raphael Meldola, who was president of both the Society of Maccabaeans and the Royal Institute of Chemistry. Remarkably, he was also awarded a second Meldola Medal in 1923, and was the only person ever to receive two medals (the rules were subsequently changed to make it impossible for an applicant to receive more than one). In 1924, he was elected to fellowship of the Royal Society and accepted the position of professor of chemistry at Leeds University, UK.

During those years, there was another important, more personal development in Ingold’s life. In 1922 he published a paper with Edith Hilda Usherwood on the specific heats of gases and, a year later, they were married. His wife was herself a talented chemist but, after a few years, she gave up her research and supported her husband’s scientific career.
At Leeds, Ingold’s interests rapidly turned to the nature of chemical bonding. Robert Robinson, who was then a professor at the University of Manchester, UK, had recognised that the current discussions, in terms of partial valency and chemical affinity, lacked any common physical basis and suggested that this could be found in the electronic theories of valency put forward by Thomson, Lewis and Langmuir.
In his 1922 paper, Robinson used Arthur Lapworth’s description of reagents as ’kationoid’ and ’anionoid’ but added what we would in modern terms call inductive and conjugative electron shifts. There are even two of the now common curly arrows to represent the movement of electrons in a conjugated system.
In his paper on tautomerism in 1926, Ingold also interpreted the results in terms of electron movements, this time making extensive use of curly arrows. The paper contains one reference to Robinson’s work published in the same year, but was the start of the long-standing controversy between Robinson and Ingold - Robinson considered that insufficient recognition had been given to his earlier ideas.
Opposing conventions
Ingold continued to develop the theory of electronic effects in organic chemistry, making a clear distinction between polarisation and polarisability effects and introducing the concept of mesomerism - equivalent to the concept of resonance between valence bond structures - as the permanent consequence of tautomeric electron displacements in a molecule before a reaction.

Ingold’s terminology differs from that of Robinson, and this is also true of his description of reagents - Ingold used the terms electrophilic and nucleophilic in place of kationoid and anionoid on the basis that whether a reagent is electron-attracting or electron-repelling is not simply dependent on its charge.
The two scientists also disagreed on the sign to be attached to substituent effects. Ingold designated the effects of electron-donating substituents as positive and those of electron-withdrawing substituents as negative. Shortly afterwards Robinson proposed the opposite sign convention. Ingold’s ideas were set out in his article in Chemical Reviews in 1934 and Robinson set out his own alternative system in two lectures published together by the Institute of Chemistry in 1932.
So, in the early 1930s, chemists were faced with two versions of an electronic theory of organic chemistry with many similar features but using a quite different terminology and sign convention. To write up their results, they had to choose one or the other, and quite quickly Ingold’s approach became the one generally accepted. One reason for the preference may have been that more people read Ingold’s article in Chemical Reviews than read the reprints of Robinson’s lectures at the Royal Institute of Chemistry. But perhaps the main reason is that much of Robinson’s interest moved from electronic theories to what he considered to be other major challenges - particularly biosynthesis - while Ingold maintained a rapid rate of publication in the area of mechanism and substituent effects. Robinson’s bitterness over the way in which his presentation of the electronic theory was supplanted by that of Ingold continued for the rest of his life. In his autobiography, Robinson maintained that the electronic theory was his most important contribution to knowledge.

In August of 1930, Ingold moved from Leeds to a professorship at University College London and remained there until his death 40 years later. In the same year, he was joined by Ted Hughes, a young postdoctoral worker who had just taken his PhD with H B Watson at Bangor. This was the start of a long collaboration which lasted until Hughes’s death 33 years later.
After arriving at University College, Ingold began kinetic and mechanistic studies. As with his studies of substituent effects, Ingold introduced a language for mechanistic studies which eventually became accepted. He first grouped organic reactions according to the underlying electronic processes involved. Thus the reactions of hydroxide ions and amines with alkyl halides together with the reactions of amines with alkyl ammonium ions were termed ’nucleophilic substitution at a saturated carbon atom’ because, although the reactions appear very different in the reagents and charges, they all involve the attack of a nucleophile and the departure of a leaving group with the bonding electrons.
Ingold made a further distinction by investigating the kinetics of these nucleophilic substitutions. He found two basic forms: in one, the rate of the reaction depended on the concentration of both the nucleophile and the organic molecule it was attacking. In the other, however, increasing the concentration of the nucleophile made no difference to the reaction rate, which only depended on the concentration of organic substrate.
As Ingold worked out, that was because in the first kinetic form - whose reaction path was termed SN2 - both incoming nucleophile and substrate molecule were involved in the slow, rate-determining step of the reaction. In the second form, SN1, the rate-determining step involved only the organic substrate, which slowly lost its leaving group, creating a carbocation that was then quickly attacked by a nucleophile.
Substitution mechanisms
Ingold described, for the first time, the two different forms of nucleophilic substitution SN1 and SN2.
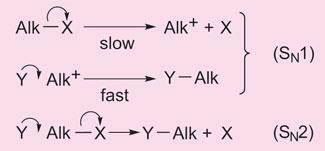
This mechanistic distinction has an important bearing on the stereochemistry of the substitution. In two series of papers, Ingold and Hughes established that the SN2 reaction, where a nucleophile attacks and a leaving group departs simultaneously, always inverts the 3D arrangement of atoms neighbouring the reaction centre - rather like an umbrella turning inside out. The SN1 reaction, however, can either invert or retain the starting arrangement, since the planar carbocation can be attacked by the nucleophile on either side. The attack on the two sides may not occur to an equal extent because of partial screening by the leaving group. This relationship of the stereochemistry to the detailed mechanism provided an explanation of a long-standing problem in organic chemistry called the Walden inversion - where if a particular substitution reaction was attempted using different conditions, it could be made to either retain or invert stereochemistry.
This mechanistic distinction has an important bearing on the stereochemistry of the substitution. A great deal of work was done on ’substitution at a saturated carbon atom’ covered in 61 papers but many other reactions were studied as well, particularly elimination, hydrolysis and esterification. This work established a language for discussing the mechanism of organic reactions in solution and was very widely accepted by other workers.
When the organic chemist William Taylor cast doubt on the evidence for the SN1 reaction, Hughes and Ingold replied with a mass of papers remarkable both for their number and for the forthright prose involved.
Later, when H C Brown, a distinguished American chemist, came to give a lecture in the anatomy theatre at UCL he was led past glass cases holding a number of skeletons of humans and apes and turned to Ingold saying, ’your earlier competitors I presume’.

The evacuation of the department to Wales during the second world war gave Ingold a number of very distracting administrative problems but, with the coming of peace, he set about rebuilding the department as a centre for what we would now term physical organic chemistry.
Three of his students from Aberystwyth (C A Bunton, Ron Gillespie and Jim Millen) joined the staff and a strong contingent arrived from Australia (David Craig, Allan Maccoll and Ron Nyholm). Peter de la Mare came from New Zealand and Kathleen Lonsdale, the crystallographer, came from the Royal Institution. Ted Hughes had been appointed to a professorship at Bangor during the war but soon returned as a professor at UCL.
Ingold quickly returned to his pre-war level of productivity, publishing 11 papers in 1947 and 31 in 1948. Much of the work in the department during and after the war concerned the mechanism of nitration. This was studied using all of the physical methods available: kinetics, cryoscopy, and spectroscopy. Studies of substitution and elimination reactions were continued, a great deal of work was done on rearrangements, and there were further incursions into spectroscopy.
In the years during and after the war the importance of Ingold’s work became increasingly recognised. This was helped by the first edition of his book Structure and mechanism in organic chemistry and it led to a number of medals, awards and honorary degrees. In 1958, he was knighted.
Ingold officially retired in 1960 but remained in the department as an emeritus professor and special lecturer. He remained very active in those years after retirement with a large number of publications on the benzidine rearrangement and electrophilic substitution at a saturated carbon atom.
Perhaps his two most important publications during this time were the definitive article (with R S Cahn and V Prelog) on the specification of molecular chirality and the second edition of his book. In spite of some health problems, he undertook a number of lecture tours abroad including the US, Australia, Israel, and Japan.
In 1970, he appeared to have a minor stroke during a lecture in Venice but was able to complete his talk. Afterwards, however, his health deteriorated markedly and on 8 December 1970 he died.
Beyond the science, Ingold was a kind and courteous man. In the 1930s he worked with Donnan to bring Jewish refugees to England. His son Keith recalls a number of occasions when he had to give up his bedroom to make way for a sudden arrival.
As an undergraduate, I remember attending a lecture by Ingold, during which a young female student arrived a few minutes late. Ingold stopped, waited for her to settle herself, and then said, ’what I have just dealt with is rather important and so I should perhaps start again’. And he did.
It seems appropriate to finish with the words with which Charles Shoppee ended his obituary of Ingold: ’He had extraordinary imagination, insight, initiative and ingenuity, he possessed one of the greatest intellects in chemistry in the 20th century, and he added a new dimension to organic chemistry.’
John H Ridd is an emeritus professor at University College London, UK
Further Reading
-
K T Leffek, Sir Christopher Ingold, a major prophet of organic chemistry. Victoria, Canada: Nova Lion Press: 1996
-
C K Ingold, Chem. Rev., 1934, 15, 225
-
R Robinson, Outline of an electrochemical (electronic) theory of the course of organic reactions, Institute of Chemistry, London, 1932
-
R Robinson, Memoirs of a minor prophet, Amsterdam: Elsevier, 1976, p184
-
C K Ingold, Structure and mechanism in organic chemistry, London: G Bell and Sons, 1953
-
C W Shoppee, Biogr. Mem. Fellows R. Soc., 1972, 18, 349












No comments yet