From the observation that plants replenish oxygen in the air to artificial photosynthesis for making liquid fuel, Mike Sutton tells the story of the chemists fascinated by chlorophyll
We are taught as young children that while humans breathe in oxygen and breathe out carbon dioxide, plants are helpfully mopping up the carbon dioxide and pumping out fresh oxygen. A little later, we discover that these mutually supportive processes are driven by two surprisingly similar molecules – haemoglobin and chlorophyll.

The story of scientists’ attempts to decipher chlorophyll’s structure and biological role is littered with Nobel laureates, and 2015 marks both the centenary of Richard Willstätter’s Nobel prize in chemistry for establishing chlorophyll’s composition and the 50th anniversary of Robert Woodward being similarly honoured for accomplishing its synthesis.
To appreciate the significance of those achievements we need to begin the story a little earlier. Several 18th century chemists and botanists – including the Englishmen Stephen Hales and Joseph Priestley, the Swede Carl Scheele, Jan Ingenhousz of the Netherlands and the Swiss Jean Senebier – observed that when green plants are exposed to sunlight, they replenish the portion of the atmosphere that sustains animal life.
At the start of the 19th century, the general acceptance of Antoine Lavoisier’s oxygen theory of combustion focused further attention on the process. Quantitative experiments by another Swiss, Nicolas-Théodore de Saussure, demonstrated that growing plants derived their carbon from atmospheric carbon dioxide – but how they did so remained mysterious.
Early extraction attempts
In 1817 the French chemists Pierre Pelletier and Jean Caventou called the green pigment they extracted from leaves chlorophyll – a name derived from the classical Greek words for green and leaf. During the next few decades, leading chemists including Jöns Jakob Berzelius in Sweden and Englishman Edward Schunk attempted to purify and analyse this substance (along with other colourful vegetable products).
They had only limited success, but meanwhile there was progress in other areas. Using an improved microscope, the German plant physiologist Julius von Sachs discovered chloroplasts – the tiny chlorophyll-secreting bodies inside plant cells – and the Anglo–Irish physicist George Stokes mapped the prominent bands of chlorophyll’s absorption spectrum.
By the start of the 20th century, it was generally accepted that plants used chlorophyll to capture the solar energy necessary to convert carbon dioxide and water into sugars and starches. This process, known from the mid-1890s as photosynthesis, is conveniently represented by a simplified equation:
6CO2 + 6H2 O ? C6 H12 O6 + 6O2
Deciphering chlorophyll’s composition
However, it was clear that something far more complex than this was going on inside plant cells, and when Willstätter took up the subject in 1902 much still remained to be explained.
Willstätter was born into a prosperous German-Jewish family in 1872, and earned his PhD at the University of Munich in 1894 with a thesis on alkaloids. He remained at Munich doing further research and some teaching, and was eventually appointed to a junior professorship there in 1902. In the following year he married Sophie Leser and they had two children before her premature death in 1908.
After holding senior academic posts in Zurich and Berlin, Willstätter returned to Munich in 1916 as the successor to his former mentor Adolf von Baeyer. During the first world war he declined an invitation from Fritz Haber to work on the offensive use of poison gas, but he did help to develop a gas mask that saved many lives.
Willstätter resigned his chair in 1924 as a protest against the antisemitism then becoming apparent in the university and he continued his research privately in collaboration with a former student. By 1938 the Gestapo were harassing him, and in March 1939 he escaped to Switzerland where he died in 1942.
It was during an earlier stay in Switzerland, between 1905 and 1912, that Willstätter did most of his research on chlorophyll. Using a variety of acids, alkalis and organic solvents, he developed techniques for separating chlorophyll from the many other substances associated with it in plant cells, and then broke it down into smaller components. Earlier investigators had noted that plant ashes contained magnesium, but he demonstrated that pure chlorophyll from an extremely wide range of sources (including pine needles and stinging nettles) always contained the same proportion – just below 3% – of that metal.
The power and versatility of the organomagnesium reagents which had recently been developed by the French chemist Victor Grignard suggested to some observers that magnesium might play a similar activating role in chlorophyll. At the time, however, too little was known about the chemistry of photosynthesis to develop this hypothesis further. Others proposed that the primary reaction might be the synthesis of formaldehyde which was then somehow polymerised to make carbohydrates. But formaldehyde could not be found in plants – and when exposed to it they usually died – so this idea also gained little traction.
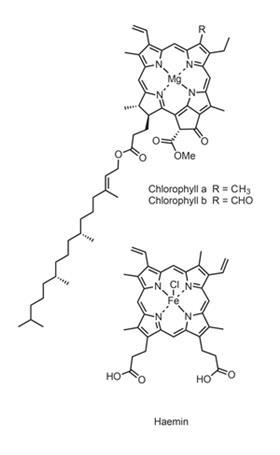
A satisfactory account of the chemistry of photosynthesis remained elusive for many more years. However, Willstätter did show that pure chlorophyll alone could not make sugars – the whole leaf was required – and he therefore argued that other agents, most probably enzymes, were involved in the process. He also proved that plants contained two slightly different variants of the chlorophyll molecule, as Stokes’ spectroscopic data had already indicated. (Several other chlorophylls have since been found in photosynthetic algae and bacteria.)
Willstätter confirmed the formulae of his chlorophyll a and chlorophyll b as C55 H72 O5 N4 Mg and C55 H70 O6 N4 Mg, and demonstrated that both molecules contained four pyrrole rings, plus a carboxyl group esterified with the long chain alcohol phytol. But the complete structure still eluded him, and by the time Willstätter received his 1915 Nobel prize (which, like several other wartime laureates, he did not collect until 1920) he had given up the search and was busy studying enzymes.
Structural confirmation
It was not until 1930 that another Munich chemist, Hans Fischer, took on the task, with encouragement from Willstätter. This was a logical move, for Willstätter had already drawn attention to the chemical similarities between chlorophyll and haemin – the red pigment of blood – while Fischer had recently gained the Nobel prize in chemistry for his haemin synthesis.
In his 1930 Nobel lecture, Fischer warned that ‘the chlorophyll molecule still presents many riddles’. Unravelling them kept his large and well organised research team busy for the remainder of the decade. They published over 100 papers on the subject, and though unable to synthesise chlorophyll, they had confirmed its molecular structure by 1940.

Successful synthesis
It was another 20 years before the synthesis of chlorophyll was finally accomplished, by Woodward’s Harvard research group. Born in Boston, US, in 1917, Woodward became fascinated by chemistry at school and studied it intensively at home.
He entered the Massachusetts Institute of Technology in 1934, and was nearly expelled the following year because he had neglected compulsory subjects which did not interest him, while pursuing advanced chemical studies on his own initiative.
Fortunately, sympathetic tutors allowed him to continue, and they were vindicated when Woodward – still only aged 20 – earned his doctorate with a thesis on the female hormone oestrone. After a year’s postdoctoral research at Harvard, Woodward joined its faculty and settled there for the rest of his professional life. Several of his investigations had commercially significant outcomes and were supported by large corporations like Polaroid, Pfizer and Ciba–Geigy (now Novartis). But while profiting considerably from these associations, he never abandoned the laboratory for the boardroom and remained one of the leading academic chemists of his era.
Reminiscences by friends and colleagues depict him as a likeable but relentlessly focused man. When a newly arrived British student enquired about holidays, Woodward replied: ‘well, I take Christmas day off’. His research teams worked hard, with long days in the laboratory frequently being followed by late night brainstorming seminars. On the positive side, however, participation in the synthesis of a ‘Woodward molecule’ often became the passport to a distinguished career.
Even allowing for the support Woodward received from his loyal legion of helpers, his personal output was phenomenal. He normally slept less than four hours per night, and while tobacco and alcohol helped to relieve the stresses of his punishing schedule, the workload seems to have taken a toll – both his marriages ended in divorce, and he died aged only 62. Chlorophyll was but one of his many notable syntheses, which also included quinine, cholesterol, cortisone, strychnine, reserpine, cephalosporin and vitamin B12.
The chemistry of photosynthesis remained elusive for years
All these achievements were greatly assisted by Woodward’s encyclopaedic memory – from which he could often summon obscure but relevant reactions at critical moments – and by his willingness to adopt new physical investigative techniques like nuclear magnetic resonance spectroscopy. However, Woodward’s most radical innovation was probably his application of molecular orbital theory to predict optimal routes for organic syntheses.
The molecules he pursued usually contained several asymmetric carbon atoms, but often only one of their numerous possible stereoisomers was biologically active. The ability to design a stereospecific synthesis from first principles could therefore save much time and effort. This predictive technique was eventually formalised in the Woodward–Hoffmann rules, published in 1965. US chemist Roald Hoffmann’s contribution to these rules earned him a Nobel prize in chemistry in 1981, which Woodward would almost certainly have shared had he not died in 1979. (Nobel awards are not given posthumously, unless the recipient dies between the announcement and the presentation ceremony.)
All of Woodward’s practical and theoretical talents were needed to navigate a path through the labyrinth of the chlorophyll synthesis, which involved 55 separate chemical reactions. After four years of intensive effort (supported by a dozen postdoctoral researchers) he completed the task in 1960, and received his Nobel prize in 1965.
Revealing chlorophyll’s role
Meanwhile, another research team in Berkeley, California, was applying equally innovative techniques to explore the role of chlorophyll in plant metabolism. Its leader, Melvin Calvin, had worked on the nuclear bomb project during the second world war – an experience that prepared the way for his peacetime use of radioactive isotopes to trace the progress of chemical reactions.

Calvin initiated photosynthesis by exposing green algae to sunlight in the presence of carbon–14 labelled carbon dioxide, and then halted the process at different stages by dumping the mixture into hot alcohol. Chromatographic separation and radiological testing of the resulting products revealed how far along the reaction chain the carbon-14 atoms had travelled.
In time it became clear that the primary function of chlorophyll molecules (in close association with specific proteins inside the chloroplasts) is to provide the energy for photosynthesis. They do so by absorbing solar photons – an operation facilitated by the many conjugated double bonds in chlorophyll’s porphyrin structure. This captured sunlight is then invested in the synthesis of energy-rich molecules like ATP (adenosine triphosphate) and NADP (nicotinamide adenine dinucleotide phosphate). These, in turn, power a sequence of light-independent reactions. The first stable product is glyceraldehyde 3-phosphate, from which larger carbohydrate molecules are then synthesised inside the plant.
Calvin and his assistants (notably Andrew Bassham and James Benson) unravelled the process in a series of classic papers published in the 1950s, and though it was Calvin alone who received the Nobel prize for chemistry in 1961, today this sequence of reactions is often called the Calvin–Benson–Bassham cycle.
Today, possible alternatives to photosynthesis offer chemists challenges and opportunities very different from those faced by Willstätter a century ago, or by Calvin and Woodward more recently. For example, at Columbia University’s Earth Institute in the US, researchers are trying to produce ‘artificial trees’ to mop up the carbon dioxide generated by our relentless consumption of fossil fuels.

Elsewhere in the US, California’s Joint Centre for Artificial Photosynthesis is currently pioneering new techniques for using solar energy to liberate hydrogen from water. But hydrogen – even in a sophisticated fuel cell – is not an ideal source of portable energy, so the centre’s ultimate goal is to develop a commercially viable solar powered process for making liquid fuel from water and atmospheric carbon dioxide.
Yet even if this ambitious target proves to be attainable, humanity will still depend (for the foreseeable future) on photosynthesis powered by chlorophyll to produce the oxygen we breathe and the carbohydrates we eat. Bearing this in mind, perhaps we should take better care of our planet’s plant resources than we appear to be doing at present.












No comments yet