Corrosion is an age-old problem, but some of the latest solutions are distinctly 21st century.
Corrosion has been a problem for as long as human have used metals – it’s no coincidence that the metals we consider most precious are those that do not corrode. Protecting metals that do corrode has involved various surface treatments, from simply painting the surface to current magnesium iron phosphate ceramic coatings for steel. Chemicals that could slow down corrosion, known as corrosion inhibitors, have been widely used in paints since the industrial revolution. But over the past decade, new types of nanostructured protective coatings have been developed that provide better corrosion protection – some delivering corrosion inhibitor only when needed, others able to heal themselves and remove flaws vulnerable to corrosion. Using clever chemistry, corrosion scientists are finding greener and longer-lasting solutions.
Today, corrosion still has a massive economic impact. The global cost of corrosion is estimated to be US$2.5 trillion (£1.9 trillion), which is equivalent to 3.4% of global GDP. Corrosion impacts on infrastructure, transport and major sectors like the oil and gas industry, who will spend millions on corrosion monitoring and maintenance activities. So why is corrosion still a problem? With all our ingenuity, why haven’t we found a solution? The answer is thermodynamics. ‘There is no foolproof solution to the problem,’ explains Mikhail Zheludkevich from the Helmholtz Centre for Materials and Coastal Research in Geesthacht, Germany. ‘All the structural metals are thermodynamically unstable and are oxidised, which causes corrosion. The only possibility is to delay the process.’
Slowing down corrosion can be achieved to some extent, and most metals used in construction will have coatings containing corrosion-inhibiting chemicals. Some of the most successful once contained chromium(vi) compounds, which form a passivating chromium oxyhydroxide layer. ‘We thought we had this figured out, but then it turned out that chromium is highly carcinogenic, so in the last 20 years or so we have had to go back to the drawing board,’ explains chemist Sarbajit Banerjee from Texas A&M University in the US. Chromium(vi) compounds are now banned in Europe and the US.
Finding the chemicals that best inhibit corrosion is still largely a trial and error process. The basic principles are to stop the cathodic or anodic half reactions occurring by impeding electron or ion transport or by creating a barrier to oxygen or water diffusion. But there is an environmental concern: inhibitors, when added to coatings, tend to leach out both on land and in the oceans, where the oil and gas industry have thousands of miles of pipeline to protect.
Graphene to the rescue?
As well as the need for greener anti-corrosion coatings, in recent years industry has introduced new alloys to replace steel, often containing lighter metals. These present new corrosion challenges. For example, magnesium added to alloys can be problematic, explains electrochemist Alicja Stankiewicz from Edinburgh Napier University in Scotland. ‘[Magnesium can] accelerate the corrosion rate because a microgalvanic couple is created between this alpha magnesium matrix phase and the other secondary phases in magnesium alloys.’ New composite materials such as those now used in aircraft also pose corrosion problems, as joining different types of materials in one structure can also create galvanic effects that accelerate corrosion.

One much talked about material that could provide a solution is graphene. ‘Graphene itself is not very effective in terms of endowing corrosion protection,’ says Banerjee. This is because it’s difficult to form pinhole-free films on metal surfaces that can provide continuous protection. ‘But blended within a polymer composite, graphene serves very well as a barrier,’ he adds.
Banerjee has made composites containing both carbon nanotubes and exfoliated graphene a few layers thick. These are dispersed in solvent and mixed with polyamic acid. The polymer then undergoes imidisation (cyclisation to create polyimide) to form a strong and rigid polyetherimide (PEI) composite coating. Systems containing 20% graphene by weight showed three orders of magnitude less corrosion than uncoated steel and one order of magnitude less than PEI alone.1 ‘Fundamentally graphene provides an excellent barrier – it literally doesn’t let anything through. On the one hand it prevents the transport of ions to the metal surface and on the other hand it is also impermeable to oxygen and prevents it reaching the surface,’ explains Banerjee. But there is a lot of work to be done before graphene is commercially used in this way .
Smart active coatings
Another approach that has gained traction is to store inhibitors in reservoirs within a coating, only to be released when corrosion is initiated or the surface is disrupted through delamination or a scratch. This gets around the problem of the inhibitor leaching away, which both reduces its ability to protect the metal and causes environmental damage. It also means smaller amounts of inhibitor can be used in coatings than previously – perhaps only 2–5% by weight.

Designing nanocontainers that can do this has been one of the most challenging aspect of creating such active coatings. They need to be the right size, have adequate loading capacity, have an affinity for the coating in which they are embedded and keep active inhibitor molecules trapped until a specific stimulus releases them.
Different research teams have used a variety of methods and materials, creating capsules of sizes varying from 100nm to 50μm. One way to make the containers is the layer-by-layer approach, which deposits alternately charged polyionic layers around a template nanoparticle, such as silica. This can eventually be dissolved out leaving the capsule. The surface properties of the capsules are also controlled. ‘That is quite an important step,’ says Dmitry Shchukin, a materials chemist at the University of Liverpool in the UK. ‘We make surface modifications to our nanocontainers – we can make them hydrophilic or hydrophobic and change the surface to negative or positive, to get the nanocontainer [distrubuted] into a paint formulation.’
‘I would say 90% of coatings we developed have nanocontainers sensitive to the pH,’ says Shchukin, who has been developing these capsules for over a decade. ‘When there is a scratch [in a coating] and corrosion starts, the metal changes its local pH and then inhibitor is released from the nanocontainers inside the coating.’ The systems are designed to be reversible, so capsules can close up again when corrosion stops.
Daniel Crespy at the Vidyasirimedhi Institute of Science and Technology in Thailand and the Max Planck Institute for Polymer Research in Mainz, Germany, has created polymer capsules from oil–water emulsions. The monomer is emulsified and phase separation inside the droplets creates the polymer shell with a liquid core that contains a corrosion inhibitor.
Polymers provide a thicker and more rigid capsule, but can work on a similar principle. ‘The polymer will swell if it is protonated, so at a lower pH value the polymer will swell and this allows the encapsulated corrosion inhibitor to be released,’ he explains.
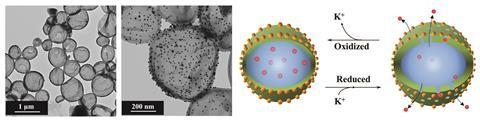
Crespy, together with Michael Rohwerder from the Max Planck Institute for Iron Research in Germany, has also created polymer capsules that are controlled by redox chemistry, rather than pH. ‘You can activate a redox-responsive polymer by the corrosion process itself,’ he says. He has made capsules from conducting polymer, polyaniline (PANI) filled with 3-nitrosalicyclic acid – a very efficient zinc corrosion inhibitor. The capsules were used in a polyvinylbutyral composite coating. When the coating became delaminated from the underlying metal, there is a decrease in potential and the PANI went from impermeable to permeable, releasing the inhibitor molecules to provide corrosion protection. The change in the capsule’s polymer structure is linked to the PANI oxidation state. In its oxidised state, intermolecular hydrogen bonding causes the polymer chains to become compact, but on reduction these bonds weaken and the loose polymer chains became permeable.2
Chloride control
Capsules are not limited to polymers and other soft materials – mesoporous materials like silica have also been used in oxide coatings. Mesoporous silica is ideal because it has a large pore volume and can incorporate inhibitors up to 40% by weight. Inorganic capsules are often smaller – less than 200nm – which allows them to be used in thin coatings. The problem is how to control the inhibitor release. One method Shchukin used was to create nanovalves, by functionalising the silica surface with large pH-sensitive weak polyelectrolytes or other polymers that can provide similar controlled release to previous polymer capsules.
Another idea Zheludkevich came up with was to create capsules that responded to chloride concentration. ‘Chloride is one of the main corrosive anions present in the environment and it’s dangerous for metallic structures,’ he explains. ‘We can design a container which will release inhibitor only when there is a high concentration of chloride ions in the environment.’ Chloride ions play a key role in stimulating corrosion, including very damaging pitting – the formation of deep holes which become acidic and encourage further corrosion. So in some environments a chloride responsive system could be valuable; for example, in or near the sea or for the automotive industry. Salt used for de-icing roads can cause problematic corrosion in cars.
Chloride is one of the main corrosive anions present in the environment and it’s dangerous for metallic structures
The materials used for these capsules are layered double hydroxides (LDH). They are made up of stacks of positively charged metal hydroxide layers with intercalated anions and solvent molecules. ‘Such compounds often have very high ion-exchange capability,’ says Zheludkevich. LDH nanoparticles are synthesised with inhibitor anions added between the hydroxide layers by ion exchange. ‘This type of container will not release inhibitor in water and will only release [inhibitor ions] where there is a high concentration of chloride ions, so you have this kind of trigger,’ he adds.
Both inorganic and organic inhibitors can be used, as long as they form anions. Chloride ions have a higher affinity to the layered double hydroxide, so will displace inhibitor anions when present. Zheludkevich has made this type of nanocapsule for protecting aluminium alloys, using a mixed magnesium and aluminium hydroxide with divanadate (V2O74-) anions as the corrosion inhibitor. These capsules were added to an epoxy primer layer at 10% by weight, and it showed significant improvements in corrosion protection over primers without these nanoparticles.3 Zheludkevich has successfully used the sample type of capsules in oxide coatings. He is now looking for greener inhibitors to replace the vanadium, but they need to have a similar charge/size ratio.
Coating, heal thyself?
But what happens when the inhibitor inside the capsules runs out? ‘One of the limitations is that you do not have unlimited encapsulated amounts,’ says Crespy. Adding more capsules doesn’t work either as this will adversely affect the mechanical and thermal properties of a coating. Rather than plugging the gap with an inhibitor, could a coating heal itself?
In self-healing coatings, the nanocapsules are able to deliver the reactants needed to reform a coating. ‘Active coatings are like putting a band aid on the crack – it doesn’t actually heal the coating entirely,’ says Banerjee. ‘Whereas a self-healing coating will have monomers of the polymer incorporated, and once there is a crack the monomer is released and it will literally stitch up the coating with new polymer.’
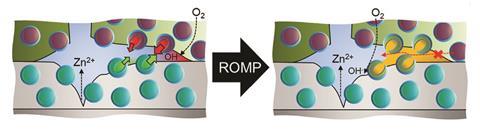
There are different methods to achieve this, but Crespy and Rohwerder have designed a system to protect zinc alloys, where capsules containing dicyclopentadiene (DCPD) were added to a zinc metal coating. On top of this is a polymer layer containing capsules filled with a Grubb’s catalyst – a ruthenium carbene complex that catalyses the breaking and making of carbon double bonds. When cracks or dents occur in these coatings and corrosion is initiated, the pH drops and both types of polymer capsules open, initiating ring opening metathesis polymerisation (ROMP). This creates a highly cross-linked poly-DCPD network that fills up and permanently heals the coating, stopping further corrosion in its tracks.4
Stankiewicz has been working on a similar concept with microgel capsules (made from the polysaccharide sodium alginate), but these will protect nickel alloys. She has co-deposited these 5μm capsules in a surface layer created through a chemical plating method, where nickel cations react with sodium hypophosphite to form a hard, corrosion-resistant nickel–phosphorus coating. The nickel surface itself acts as a catalyst for this process. But if scratched and vulnerable to corrosion, the capsules come into play and repair the coating. ‘In a corrosive medium, microgels release both chemicals, which can react with each other and create the coating,’ says Stankiewicz. ‘Because the nickel matrix itself is a catalyst of nickel deposition, the reduction process of nickel [and new coating deposition] occurs at the coating failure sites.’
Testing times
Self-healing and active coatings are now well-established in academic labs, but have they yet made their way to industry? The paint and coatings industries tend to keep quiet about their exact formulations, including methods for stopping corrosion. ‘I know that there are several companies that claim to produce [self-healing] coatings,’ says Shchukin. ‘Industry is quite conservative, especially the car industry.’ This is not surprising given what is financially at stake if a material fails. So how can they be sure a new self-healing coating will successfully prevent corrosion?
They paint several cars and put them in places like Siberia or the Sahara and wait for a couple of years
In the lab, materials and coatings are electrochemically tested to find average corrosion rates in millimetres per year. Accelerated corrosion tests over weeks or months are also standard methods. Samples are exposed to cycling humidities, temperatures, chloride levels and even pressures. But according to Banerjee, ‘It’s difficult to transfer lab research into predictions of what’s actually going to happen in the field, because small failures can launch catastrophic cascades of processes that get out of control.’
Industries have their own standard tests, such as the salt spray test, where a salt fog is sprayed on a metal at a high temperature. ‘In fact this method does not have very good correlation with real conditions and there are many reports which say it has nothing to do with the real performance of a system, but still industry tries to rely on these methods, which are often misleading – it’s a problem,’ insists Zheludkevich.
The car industry often relies on relatively long-term testing of four to five years. ‘They paint several cars and put them in different places like Siberia, the Sahara Desert, or Brazil for instance and wait for a couple of years just to see what’s going on,’ says Shchukin.
Active coatings are slowly making their way to real applications. Self-healing polymer top coats have been developed by German paint manufacturers Bayer Materials Science and Dutch company AzkoNobel. Both Toyota and Nissan reportedly use such systems, although these types of coatings need to be heated to activate the self-healing properties. Zheludkevich is also commercialising his LDH smart nanocontainer coatings through Smallmatek, a company based in Portugal. Graphene-based coatings are now being pioneered by Tata Steel – their first graphene coated steel beams being launched in 2017.
Synergistic effects
Researchers are now looking at improving coating performance and one route is to look at inhibitor combinations. ‘You can get synergistic effects, where combinations [of inhibitors] work better than single inhibitors,’ says Zheludkevich. He has recently experimented with coatings combining both cerium(iii) and mercaptobenzothiazole nanocontainers in epoxy coatings for an aluminium alloy. The cerium cation and mercaptobenzothiazole anions worked better together and showed exceptional corrosion protection.5
Banerjee is also working on combining other approaches with graphene coatings. ‘The [graphene] nanocomposite is quite versatile because it allows you to mix and match the component as required for a specific coating application.’ He has added magnesium particles as an electroactive coating that provides sacrificial cathodic protection. On exposure to saline environments these coatings performed better than standard zinc-rich primer coatings. The magnesium is preferentially oxidised and its corrosion products provide passivation.6
The size of the impact that nanostuctured self-healing and stimuli-responsive coatings will have in reducing corrosion remains to be seen. Researchers are continuing to improve the properties and performance of these coatings, and the number of potential combinations of materials and methods is still growing. So far, from a materials chemistry perspective, we have just scratched the surface – pun intended.
Rachel Brazil is a science writer based in London, UK
References
1 R V Dennis et al, Am. Ceram. Soc. Bull., 2013, 92, 18 (https://bit.ly/2khHuE0)
2 A Vimalanandan et al, Adv. Mater., 2013, 25, 6980 (DOI: 10.1002/adma.201302989)
3 M L Zheludkevich et al, Corr. Sci., 2009, 52, 602 (DOI: 10.1016/j.corsci.2009.10.020)
4 A Vimalanandan et al, Adv. Mater., 2015, 27, 3825 (DOI: 10.1002/adma.201501044)
5 M A Zadeh et al, Corr. Sci., 2018, 135, 35 (DOI: 10.1016/j.corsci.2018.02.018)
6 R V Dennis et al, Ind. Eng. Chem. Res., 2014, 53, 18873 (DOI: 10.1021/ie500897n)












1 Reader's comment