Nina Notman speaks to the researchers unpicking the many ways the climate crisis is impacting our oceans – and vice versa
The oceans are the superheroes of anthropogenic climate change. They cover 71% of the Earth’s surface but have absorbed 90% of the excess heat caused by human emissions of greenhouse gases. Additionally, the oceans have limited excess heat by sucking up a quarter of anthropogenic carbon dioxide. In a nutshell, without the oceans’ outsized effects, the Earth’s surface would be significantly warmer than it is today.
But this vital natural buffering role is not without its consequences. Warmer oceans, for example, are causing rising sea levels that threaten island nations and coastal communities. Excess carbon dioxide is also changing the chemistry of the oceans. The combined effect of this, along with warmer waters, is harming marine ecosystems and affecting seafood populations that provide food and employment for communities. There is also concern that the oceans’ heat buffering effects may not keep pace with an escalating climate crisis.
Warming oceans
Over the past 30 years, the amount of warming has been equivalent to the 8 billion people on the planet continuously dumping 34 kettles full of boiling water into the oceans, explains Gregory Johnson, a physical oceanographer at the US National Oceanic and Atmospheric Administration (NOAA) in Seattle. As a result, our oceans are warming at the average rate of 0.2°C per decade at the sea surface. In addition to harming marine organisms, warmer oceans are slowing down the global ocean circulation system – the ocean conveyor belt. ‘The whole ocean is moving around very slowly, all the time,’ explains Stephanie Henson, a marine biogeochemist at the UK’s National Oceanography Centre (NOC) in Southampton. A single water molecule takes, on average, 1000 years to travel a complete loop through the global oceans and back to where it started.
The whole ocean is moving around very slowly, all the time
A vital step in the ocean conveyor belt is for the water in the upper ocean to sink to the deep oceans where it then joins the deep ocean currents. But because climate change has mostly warmed the oceans’ upper layers it has made the water near the top warmer and therefore more buoyant, changing the temperature gradients in the water column and slowing down this crucial sinking process.
The slowing of the ocean conveyor belt is worrying for a host of reasons, including due to its vital role in distributing heat around the globe. Significant changes to these major ocean currents are widely predicted to have catastrophic effects on weather and climate. For Western and Northern Europe, the slowing down – or even collapse – of the Atlantic Meridional Overturning Circulation, the current that includes the Gulf Stream, is currently of huge concern.
The solubility pump
Oceans don’t just move around heat. Changes to ocean circulation also impact the distribution of nutrients and oxygen needed by marine ecosystems to survive. In addition, the processes through which oceans absorb carbon dioxide from the atmosphere are affected. Two distinct mechanisms sequester carbon into the deep oceans: the solubility pump and the biological carbon pump.
The solubility pump is a chemical process that starts with carbon dioxide from the atmosphere dissolving in the upper ocean. Some carbon dioxide returns to the atmosphere quickly, while some is turned into carbon species that are transported into the deep ocean by natural ocean circulation. Once in the deep ocean, this carbon can remain locked away for centuries depending on which ocean currents it hitches a ride on.
We know the thermodynamics of carbon dioxide in seawater pretty well
As the solubility pump depends on ocean circulation, slowing down ocean currents slows down this pump. Warmer oceans also harm the pump in a second way – by reducing the amount of carbon dioxide that can be absorbed per unit of water. This is because carbon dioxide is less soluble in warm water.
Despite this double attack on its efficiency, the solubility pump is currently still performing well. But it is projected that if ocean temperatures continue to rise, it may struggle to keep pace. ‘We know the thermodynamics of carbon dioxide in seawater pretty well [and] we can calculate how much less carbon dioxide would be stored in the ocean per degree of warming of the ocean,’ says Francois Primeau, a physical oceanographer at the University of California, Irvine, in the US. But how changes to ocean circulation will impact the solubility pump is less well understood.
Ocean chemistry changes
Despite being a positive from a climate perspective, the increase in solubility pump since the industrial revolution is having a negative impact on some marine ecosystems. This is due to changes it causes to the carbonate chemistry and pH in the oceans – changes which are collectively coined ocean acidification.
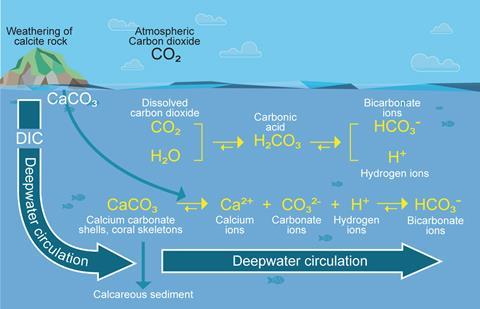
When carbon dioxide dissolves in water it produces carbonic acid which then dissociates to produce hydrogen ions. ‘The pH [of the ocean] has declined on average by approximately 0.1 [pH units] since the industrial revolution,’ says Dwight Gledhill, a chemical oceanographer at the NOAA in Maryland. Because the pH scale is logarithmic, this seemingly small change equates to a 30% increase in acidity. In more acidic oceans, young shell-producing marine organisms need more energy to survive and, as a result, may not have enough energy reserves to develop essential self-protection tools such as more durable shells.
When carbonic acid dissociates, it also triggers a cascade of reactions that alters the balance of other dissolved carbon species. This includes a reduction in carbonate ion concentration. ‘The proton combines with a carbonate ion to make bicarbonate, which means that you’re losing carbonate concentration over time,’ says Gledhill. An ocean supersaturated in carbonate is crucial for crustaceans, molluscs, and a host of other marine species to form shells efficiently.
Over aeons of time natural buffering generally keeps large changes in ocean pH in check against changes in carbon dioxide, explains Gledhill. This includes upping the weathering of carbonate rock so more calcium carbonate flows into oceans when atmospheric carbon dioxide levels are high. Nature’s buffering processes have not been able to keep up with the speed of carbon dioxide additions to the ocean since the industrial revolution. ‘We’ve popped the cork off the geologic reservoir,’ Gledhill says. ‘The changes that have been observed [in recent years] are at least 10 times faster than any period over the past 50 million years.’
Biological carbon pump
Marine life plays a starring role in the oceans’ second tool for sequestering carbon: the biological carbon pump. In the first step of this system, microscopic photosynthesising plant-like organisms that live on the surface of the ocean, called phytoplankton, take up carbon dioxide and convert it into oxygen and carbon-based molecules. Next, phytoplankton are eaten by microscopic animals called zooplankton. ‘Once [the zooplankton have] eaten, they poop. Those faecal pellets are quite dense, and full of carbon, and they sink down into the ocean,’ says Henson. Carbon also makes it to the deep ocean when phytoplankton die and clump together, making aggregates heavy enough to sink to deep enough depths for the carbon in their bodies to be sequestered.
Marine snow is a collective term for all these sinking carbon particles. Since marine snow can sink much faster than carbon species are moved downward by ocean circulation in the solubility pump, the biological carbon pump is the fastest method of transporting carbon from the ocean surface to the deep ocean currents.
There are however many more open questions about the biological carbon pump compared to the solubility pump. This makes it challenging to predict how it might respond in an escalating climate crisis. Warmer and more acidic oceans will affect both the species and abundance of phytoplankton in many locations, but whether this will make the pump more or less efficient isn’t clear. ‘We don’t have a good handle on how this is going to ultimately change how much carbon is stored by biology in the ocean,’ says Henson. ‘The IPCC, the Intergovernmental Panel on Climate Change, identified the lack of understanding of the biological carbon pump as one of their key knowledge gaps in the latest report,’ she adds.
Data demands
Before the future can be projected, a better understanding of how the oceans work now is needed. The current predictive models are ‘super uncertain’, Primeau says. To build these improved models will require much more observational data.

The first large-scale scientific exploration of the oceans was the Challenger Expedition, which sailed from 1872 to 1876. The scientists on board travelled nearly 80,000 miles collecting water samples and data on ocean temperatures, currents and marine organisms. Today, oceanography still relies heavily on ship expeditions but ship data is also supplemented with information from a myriad of other sources. These include fixed-point ocean observatories, robotic floats and gliders, and satellites.
The Global Ocean Ship-based Hydrographic Investigations Program (Go-Ship) is a key source of high-quality oceanographic information. Through Go-Ship and its predecessors, researchers from 19 different countries have been touring the global oceans since the mid-1970s. The cruises collect oceanographic data and water samples approximately every 30 miles along fixed paths, with each path revisited once a decade. ‘We’re just beginning our fourth decadal survey,’ says Elaine McDonagh, a physical oceanographer at Norce (the Norwegian Research Centre) in Bergen and NOC in Southampton. She is also the co-chair of the International Go-Ship programme and science committee.
Go-Ship cruises observe water temperature, pH and the concentrations of oxygen, various nutrients and inorganic carbon species. The scientists on board also look for tracers such as chlorofluorocarbons (CFCs) in the water samples. The age and amount of CFCs in the water column allows ocean circulation rates and pathways to be calculated.
In 2016, a Go-Ship pilot bolt-on called Bio-Go-Ship launched. ‘Bio-Go-Ship is considering marine ecosystems and ocean health through observations of plankton communities,’ says McDonagh. By observing the biology alongside the physical and chemical properties, it is easier to unravel the links between them. ‘The whole becomes much greater than the sum of the parts,’ she explains. There have been 13 Bio-Go-Ship expeditions so far. Three are planned for 2024 and the end goal is to collect biological data on all the Go-Ship cruises.
Fixed-point ocean observatories
Go-Ship cruises provide once-a-decade, high-resolution snapshots of the oceans. Fixed-point ocean observatories, meanwhile, collect high-frequency, high-resolution information about how the ocean is changing in specific locations over many years and even decades. This data complements ship measurements, explains Richard Lampitt, an observational biogeochemist at NOC in Southampton.

There are hundreds of fixed-point ocean observatories around the globe, including on the coasts and in the middle of the oceans. These observatories have cables that stretch from a buoy on the ocean surface to the seafloor with automatic sensors and samplers attached to the cables at various depths. An increasing number of observatories also collect information useful for understanding the biological carbon pump.
Lampitt is responsible for the 4850m deep Porcupine Abyssal Plain Sustained Observatory, which lies about 500km southwest of Ireland in the Northeast Atlantic. ‘It’s the most sophisticated [fixed-point ocean observatory] in the world,’ he says. It has been operating consistently since the late 1980s. Its 6m high buoy belongs to the Met Office and collects meteorological data, such as air pressure and temperature, humidity, and wind speed and direction. Sensors in the upper ocean include those for determining sea temperature, pH, salinity, concentrations of nutrients and oxygen, and chlorophyll fluorescence. The latter is a proxy for phytoplankton concentrations. Cameras are installed on the seafloor to monitor marine animal abundance and diversity. All the data is transferred by satellite to shore. ‘Every few hours, we get more data coming on the status of the ocean at that moment,’ says Lampitt.
There’s snow secrets
Lampitt and his team also collect samples of marine snow from 3000m below the surface at the Porcupine Abyssal Plain Sustained Observatory. This allows them to measure carbon sequestration flux – the rate at which carbon particles fall deep enough into the ocean that they are sequestered for 100 years or more. They use sediment traps, large funnels that concentrate particles into bottles on a carousel that rotates every two weeks, to collect the marine snow. Approximately once a year, the scientists visit the observatory by ship to return the samples to shore for analysis. ‘From the point of view of climate change, the crucial measurements are organic carbon and inorganic carbon,’ says Lampitt. Other routine measurements on the marine snow include concentrations of the nutrients nitrogen, phosphate and silicate.

The scientists can also probe the marine snow in more detail. They can, for example, isolate DNA from individual particles and bulk marine snow. This genetic information helps build up knowledge of the marine organisms involved in the biological carbon pump and therefore build more accurate models. The concentrations of thorium-234 can also be measured, as a proxy for organic carbon sequestration rates. Thorium-234 is a naturally occurring sticky radioisotope with a half-life of 24 days, explains Ken Buesseler, a marine chemist at Woods Hole Oceanographic Institution in Massachusetts, US. It is absorbed from the atmosphere and then sticks to the carbon particles in the ocean. The less thorium-234 on a carbon particle, the longer it has taken to sink down from the upper ocean.
Some research cruises also use sediment traps to collect marine snow. At any one time tens of research vessels (beyond those involved in Go-Ship) are out on the open seas and travelling along the coastlines collecting oceanographic data. The Nasa Export Processes in the Ocean from Remote Sensing (Exports ) programme, for example, is a key current effort aimed at studying the biological carbon pump. Through this programme, Buesseler and his co-workers are exploring how phytoplankton numbers and growth rates in the upper ocean are linked to the amount of carbon sequestered long term. Quantifying this relationship would allow carbon sequestration flux to be predicted from the continuous real-time phytoplankton information already collected using Nasa satellites.
Scientists involved in Exports visited the North Atlantic, a region with a high flux rate, and the North East Pacific, which has a slow flux rate. Buesseler sampled 60 locations, three times each, with a sediment trap array that collects samples at five depths in the top 500m of ocean. Other scientists onboard deployed underwater microscopes to take millions of digital images of phytoplankton and marine snow. ‘Underwater microscopes [can take images from the] 10µm to centimetres scales,’ says Buesseler. The images are then processed with AI to determine the phytoplankton species and the size and origin of the marine snow. The microscopes were lowered over the ships’ side and also deployed on autonomous underwater gliders.
An autonomous future
Autonomous underwater gliders can also hold a suite of sensors measuring water temperature, salinity, depth and oxygen concentrations. Some also have tools to measure chlorophyll fluorescence and optical backscattering sensors, which provide a proxy for concentrations of carbon particles. ‘We can use this [backscattering] information to derive estimates of carbon flux,’ says Henson.
The gliders are about 2m long and move up and down in the water using a mechanical pump that changes their buoyancy. They travel at less than a mile an hour through the oceans, sending data back to shore by satellite each time they surface. ‘The gliders really help us to understand changes over time, because they can be left out for months at a time and they’re continuously observing,’ Henson says.
The entire array of Argo floats uses less power than a PlayStation 2
Since 2017, Henson has been using gliders to quantify seasonal variability in the carbon flux in the twilight zone (100m to 1000m below the surface) in regions including the tropical Atlantic and Southern Ocean. Recently however she has turned her attention to another type of underwater autonomous device: Biogeochemical Argo floats.
The Biogeochemical Argo floats are a newer iteration of the Argo floats that have been roaming around below the oceans’ surface since 1999. Around 3000 of the original design of Argo floats are currently deployed around the globe. ‘They spend about nine of their 10 day cycle drifting with the ocean currents at about 1000m depth making spot measurements of temperature, salinity and pressure,’ explains Johnson, who has worked on the Argo programme since its launch. The floats then rise to the surface using a similar buoyancy mechanism to the gliders and ‘phone home’ before sinking again, he adds. The floats are typically a little over 1m high and are powered by around 12 DD lithium batteries. ‘The entire array of Argo floats uses less power than a PlayStation 2,’ says Johnson. ‘They are remarkably efficient and last on average more than four years.’
In 2014, the first new iteration of the Argo floats was deployed: the Deep Argo floats. As their name implies, these are designed to withstand deep ocean pressure, with one design going down to 6000m and another to 4000m. Currently, 190 Deep Argo floats are deployed, but the goal is to have 1200. ‘Having more would better allow us to monitor sea level budgets and really nail down Earth’s energy imbalance by knowing how much the deep ocean is warming,’ says Johnson.
The first Biogeochemical Argo float was launched in 2016. Today, more than 550 of these floats are in operation, with a goal of 1000 in the future. This third iteration of float has the same conductivity, temperature and pressure sensors as other Argo float types, plus between one and six additional sensors. ‘These sensors can measure oxygen, nitrate, pH, optical backscatter, chlorophyll fluorescence and downwelling radiation,’ Johnson says.
Together all the Argo floats are constantly providing copious valuable information on the global oceans, but it is lower quality data than that collected by research cruises. ‘The optimal observing system, which is what we’re trying to move towards, is nested,’ explains Gledhill, with the layers in the nest coming from different ocean monitoring tools.
And the more data that is collected, the better scientists will be able to predict how the oceans will respond if the climate crisis continues to escalate. But even if humanity were to suddenly pull together and act to maintain the atmospheric carbon dioxide at its current level, the oceans will still keep changing – for many years. ‘That’s because of that decadal timescale to the oceans,’ says Johnson. It takes decades for the oceans to come to equilibrium with the atmospheric conditions. ‘It takes a lot to get it moving, but once it’s moving, it takes a lot to get it to stop, and it’s moving now.’
Nina Notman is a science writer based in Salisbury, UK

















No comments yet