Do you know your cold compound from your London dry? Nina Notman sorts through the botanicals to find the perfect cocktail
We’re in the middle of a gin renaissance. The sales of this juniper-flavoured spirit are rocketing globally. The UK, for example, saw a 12% increase in volumes sold in the year up to June 2017, according to the Wine and Spirit Trade Association. It also reported a 32% increase in British gin exports in the past five years.
It is believed that ‘mother’s ruin’ was first made as a medicinal tonic in the early 17th century, and the gin and tonic was developed in the 19th century as a way to make quinine more palatable for British officers at risk of catching malaria in India. A G&T remains the most popular way to drink gin, but its use is also surging in cocktails and – like whisky – as a neat drink served over ice.
Multinational drink manufacturers are meeting much of gin’s increased demand; Bacardi’s gin brand, Bombay Sapphire, for example, has been upping its output by approximately 10% per year. But an increasing number of smaller gin producers have sprung up in the past few years to offer drinkers quirky alternatives to the big brands. This trend follows that of craft breweries and whisky distillers in recent decades.
Gin is produced by using juniper berries and other botanical ingredients to flavour an already-distilled neutral alcohol. In the case of cold compound gins, after any botanical solids have been removed, the liquid is diluted and bottled. Compound gins are rarely labelled as such due to negative connotations with the term, but many of the budget gins are made this way.
The costlier, but arguably tastier, way to make a gin is to redistill the alcohol after the botanicals have been added. This is how the small craft producers and many of the big brands make their spirits, and the gin-making method discussed in this article.
The raw materials
The neutral alcohol used for gin making is highly concentrated ethanol, which by law must be of agricultural origin. This is normally a grain, but some gin producers use ethanol from grapes or molasses, the by-product of sugar refining, instead. The ethanol is purified by repeat distillation to reach at least 96% alcohol by volume.
Many gin makers buy in the neutral alcohol rather than make it in house, explained Eli Breitburg-Smith, from the Baltimore Whiskey Company, US, as he showed me around his distillery. Pharmaceutical companies are the dominant suppliers, as they have huge stills designed to produce extremely pure ethanol for drug manufacture. Not all 96% ethanols taste the same, as the remaining 4% can make a big difference to the flavour and the mouthfeel of the end product. The Baltimore Whiskey Company uses ethanol made from corn for its gin production. This craft distiller predominantly makes rye whiskey but since it opened its doors in 2015, the same 1000 litre copper still has also been used to produce a handful of other spirits, including a gin that is also sold after ageing in barrels.
Another small-scale distiller is chemistry graduate Toby Whittaker of Whittaker’s Gin in Harrogate, UK. His distillery also started operations in 2015, and now produces 3000 bottles a month from its two 300 litre copper stills. The neutral alcohol they use is from wheat and barley. Bombay Sapphire, which produces 40 million bottles a year, favours a wheat-based neutral grain spirit from continental Europe. Bombay Sapphire houses four copper stills at its distillery in Laverstoke, UK – two 3200 litre stills and two with a 12,000 litre volume.
Apart from water and ethanol, the only other raw materials used for distilled gin-making are natural flavourings referred to as botanicals. The predominant flavour is always juniper; this is especially true for the London dry gins such as those produced by Whittaker’s Gin and Bombay Sapphire. The Baltimore Whiskey Company makes a contemporary American-style gin, meaning juniper is still the dominant botanical but with more prominent flavours from the other botanicals than in London dry gins.
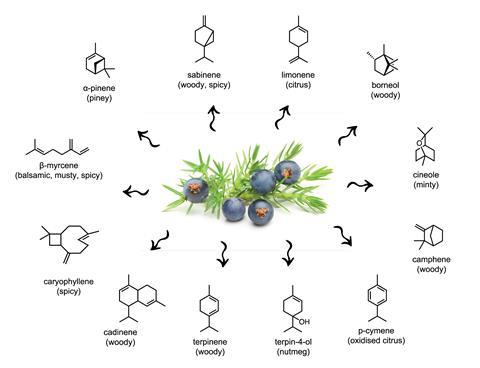
Ethanol draws many different oils out of the cells of juniper berries; these are mainly monoterpenes (C10 structures) and also some sesquiterpenes (C15 structures). C10 flavour molecules that have been identified in juniper berries include α-pinene (which gives a piney flavour), sabinene (woody, spicy), limonene (citrus), borneol (woody), β-myrcene (balsamic, musty, spicy), p-cymene (oxidised citrus), camphene (woody), cineole (minty), terpinene (woody) and terpinen-4-ol (nutmeg). The predominant C15 molecules are farnesene (with a floral note), cadinene (woody) and caryophyllene (spicy).
‘These all combine to give a piney, woody, peppery, citrusy, woody, spicy and mentholy taste,’ explains Matt Hartings, a chemistry professor at American University in Washington DC, US, who has written a book on kitchen chemistry. The exact flavour molecules, and especially their proportions, vary between juniper species and also depend upon where the juniper grew – which has an obvious knock-on effect on how the berries taste and smell. Gin producers often, therefore, have favourite farmers that they source their entire juniper (and other botanicals) supply directly from.
Taste sensation
The juniper in gin can be complemented by a wide range of other botanicals (see Signature botanicals box, below) to bring many more flavour molecules into the mix. But the taste of the end product will not just be the sum of its component parts. ‘How flavour molecules interact with your sensory system completely changes based on what they’re present with,’ explains Hartings. ‘Going from one gin to the next, you’re going to get those hints of juniper slightly differently depending on what other botanicals they’re mixed with.’
Attractions between similar flavour molecules influence how each of them interacts with the flavour receptors. ‘An interaction between two different flavour molecules, suspended in the mouth at the same time, is going to change the overall energetics of how each of those molecules individually interacts with the flavour receptor,’ explains Hartings. ‘Everything you add to that system is going to alter the final free energy of interaction of each molecule with your flavour receptors.’
But molecular attraction is not the whole story: competition between flavour molecules also plays a role. ‘You can also have two different molecules fighting for interaction with the same flavour receptor,’ Hartings says. ‘Those same molecules interacting within the water and ethanol matrix can also prevent them from interacting so strongly with your taste receptors.’ Adding other liquids, such as mixers, to your gin also changes the way it tastes for the same reasons. In tonic water, for example, quinine is attracted to a number of the flavour molecules in gin. The aggregates of flavour molecules create a taste sensation that is completely different from just gin or tonic on their own. ‘There’s a lot of different ways that you can both dampen and alter your flavour experience just by the way different molecules in your gin are interacting,’ Hartings says.
So how does a gin recipe get formulated? ‘When I think of gin making, I go back to thinking about alchemists,’ says Hartings. You can’t just start off deciding what flavour molecules you want in the final product, and buy them in a bottle, mix them together and assume you’ll get the taste you wanted, he explains. Not enough is known about the science yet to do this.
Recipe formulation is therefore largely down to trial and error with a little bit of guidance from the right people, explains Whittaker. ‘We purchased a small still and 25 litres of neutral grain spirit, and started distilling with various different berries. We would then taste and make notes on each one.’ Whittaker also wanted to include botanicals that represent the location in his distillery. ‘We incorporate hawthorn berries, bilberries, bog myrtle and garden thyme to reflect the fact that we live in Nidderdale and all these things grow in Nidderdale.’
And while distillers may be open about the botanicals they add to their gin, the exact proportions remain a tightly guarded secret. ‘Our master botanicalist is the only person that knows the recipe for all of our gins,’ explains Sam Carter, senior brand ambassador for the Bombay Spirits Company. ‘He gets all the botanicals sent to him in Geneva, he then divides them into the right ratio and sends us the boxes of botanicals ready to load into the infuser basket.’
Signature botanicals



The art of distillation
But choosing the right botanicals, with the right flavour molecules, in the right ratios isn’t the whole story, how the ethanol extracts their oils is also important for determining the final flavour of a gin.
The Baltimore Whiskey Company macerates all the botanicals in ethanol in the still at room temperature for around 12 hours before distillation. Whittaker’s Gin leave two botanicals out of their 24 hour long room-temperature macerating process, adding the lemon peel at the very end just before the distillation. ‘This means the citrus flavour doesn’t get swallowed and overcome by the other flavours – and the garden thyme we put in a gin basket,’ says Whittaker. A gin, or infuser, basket is placed in the arm of the still so that ethanol vapour passes through it during the distillation. ‘This provides a softer flavour,’ explains Whittaker. ‘A lot of distillers use both macerating and the gin basket.’
Bombay Sapphire places all its botanicals inside the gin basket, which is unusual. ‘The infuser basket is about 25 feet up in the air and 35 feet across the still house, about half a foot from the condenser,’ says Carter. ‘The key thing about our distillation is that we’re not cooking the botanicals. Many of these botanicals have natural sugars in them, so if you boil them in alcohol they tend to float and move to the sides and touch the copper, meaning the sugars in the botanicals start to cook and caramelise,’ Carter explains. ‘By vapour infusing, we’re representing the botanicals in a natural, raw way rather than a cooked way.’

The shape of the still can also affect the flavour of the gin, as the amount of internal reflux varies between different still shapes. ‘We designed ours to be a low rectification still, so it’s short and the lyne arm points down meaning there’s not much that re-condenses and falls back into the still,’ explains Baltimore’s Breitburg-Smith. This design allows the heavier oils to carry over more quickly. ‘This lends a really nice sweet character to the gin,’ he says.
By contrast, a taller still or a fractional column will give a ‘cleaner’ product, although this is more often used for whisky making than for gin. This is the same as the difference between the simple distillation and a fractional distillation of a mixture in the chemistry lab; the repeated vaporisation and condensation cycles caused by the fractionating column will result in a purer end product. Depending on the flavour profile a distiller is looking for, this may or may not be desirable.
Bombay Sapphire uses a fractionating column for its premium product, Star of Bombay. This isn’t for the reason discussed above though, as no botanicals have yet touched the ethanol at that point in their process; instead the reflux column is used to slow down the rate at which the alcohol vapour passes through the gin basket.
‘We fit a reflux column on the still, and as the alcohol vapour comes up through the middle of the column, some of it will condense and fall back down. Much less alcohol vapour manages to get through and the flow rate slows down. By the time it gets to the infuser basket, the vapour’s got longer to linger and extract more flavour from the botanicals,’ says Carter. The flow rate is adapted during the distillation run. ‘It is much slower at the beginning, we speed up a little bit in the middle, and then we slow it down again towards the end to be able to catch more or less of different botanicals because different botanicals give us their flavours at different points.’
Collecting the distillate
The volatility differences between the flavour molecules that Carter is referring to are significant enough for the gin distillate to taste and smell different throughout the distillation process. ‘I think one of the interesting things about gin making is seeing the different flavours come out throughout the run,’ explains Breitburg-Smith. ‘There’s a harsh flavour at the beginning coming out that we don’t like, so we cut out that.’ Throughout the rest of his 16h distillation, the gin flavours subtly change. ‘Then towards the end of the run, the green tea flavour becomes really prominent before getting this almost spent tea characteristic to it, and that’s where we make our cut.’ These cuttings at the beginning and end are called the heads and tails, whilst the drinkable section is the hearts.
Bombay Sapphire starts collecting the hearts, ‘as soon as our distillers smell the bright, fresh citrusy notes’, says Carter. Towards the end of the run, some sulfury notes come through. ‘The distillers know approximately where this is so they’ll be stood by the spirit safe and they’ll nose it and taste it constantly until they get to the point and they switch it to the tails.’
Not all distillers taste and smell their gin during each distillation run, although it’s a definite perk of the job! Cutting points can be determined using percentage alcohol measurements: a drop in the alcohol by volume signals the end of the hearts. Craft distillers tend to use small hydrometer for this, whilst Bombay Sapphire uses Coriolis mass flowmeters.
The final stages
Due to the different volatilities of the flavour molecules, the distillers mix together all the distillate from each gin run to ensure a consistent taste to their products. The large volumes produced by Bombay Sapphire’s stills necessitate a large-scale solution: huge nitrogen bubbles are pumped into tanks of gin to rouse them. ‘A humongous two-to-three metre wide bubble gets released into the bottom of the tank and as it’s bubbling up through the liquid it stirs it,’ says Carter. ‘This is the most efficient and the safest way of doing it because if you had a mechanical paddle you could get a spark from the motor.’ Next, around 10 different distillates from all four stills are roused together – again this ensures consistency of taste.
All three distilleries carry out their quality control by tasting and nosing. Bombay Sapphire has also recently begun using a gas chromatograph (GC). ‘A sample of gin is run through the GC. The resulting chromatogram is then mapped against our standard one just to make sure that nothing crazy is happening,’ says Carter.
The next stage is adding water to the gins to meet the required proof. Some distilleries also add sugar, alcohol or essences and other flavourings at this point, but none of the three distilleries discussed here do. The gins are now ready for bottling.
A sample of gin is run through a GC to make sure that nothing crazy is happening
Some craft distillers, including the Baltimore Whiskey Company, put some of their gin in barrels at this point in the production process. ‘We put our gin into used rye whiskey barrels for six to nine months,’ explains Breitburg-Smith. ‘It changes a lot of the botanical notes. The more volatile components like the jasmine flower and the green tea are the first things to evaporate through the barrel. So you lose the forward component a little bit. Then what is left reacts with the char and with oxygen, so you get some honey, vanilla, and really interesting characters coming through. The coriander and the juniper flavours also become a little sharper in the barrel.’
The significance of the flavour difference between the immediately bottled gin and the barrel-aged gin comes as a surprise to me. Tasting and smelling the neat gins, side-by-side, it was hard to believe they contain the exact same botanicals distilled in the exact same way. Did I have a favourite? You bet I did! And a bottle of the barrel-aged gin came home with me. Cheers!
Nina Notman is a science writer based near Baltimore, US
References
M Hartings, Chemistry in your kitchen, Royal Society of Chemistry, 2016
A Stewart, The drunken botanist, Timber Press, 2013














2 readers' comments