Can we explain the strange properties of water by thinking of it as two different liquids? Rachel Brazil dives into the ongoing debate
Water, the most commonplace of liquids, is also the strangest. It has at least 66 properties that differ from most liquids – high surface tension, high heat capacity, high melting and boiling points and low compressibility. One school of thought is that water is not a complicated liquid but ‘two simple liquids with a complicated relationship’. For some, this statement contradicts the basic principles of physical chemistry; for others it explains just why water behaves in such an anomalous way.
Over the last decade the academic arguments have reached boiling point. ‘[It’s] bringing out very strong, almost religious opinions among different scientists,’ says Anders Nilsson, a chemical physicist with appointments at Stockholm University in Sweden and Stanford University in the US. Chemists have attributed water’s strange properties to the tetrahedrally arranged hydrogen-bonding networks that it forms, but exactly what is going on, particularly when water is in a supercooled state, is still up for debate.
Like many other chemists, Nilsson assumed earlier in his career that the structure of water was well understood, but started to realise that was not the case. For the last 10 years he has concentrated on understanding water’s extraordinary behaviour and his recent work adds weight to the theory that water isn’t one liquid, but two distinct separate liquid structures that can coexist – an idea that is still controversial.
It’s a lot of hydrogen bonding for such a small molecule
‘If you look at the simple thermodynamic and kinetic properties of liquids, as you change pressure and temperature, they all behave the same way,’ explains Nilsson. As you cool a liquid, its density increases, and its heat capacity and compressibility decrease. ‘Almost all liquids on the planet behave like that. Except for water.’ This strangeness comes into focus as water is cooled to 4°C, where its density reaches a maximum, below which it starts to decrease again.
The explanation chemists are taught is that while most liquids are disordered, with their molecules constantly rearranging, water differs due to its network of hydrogen bonds. Hydrogen bonds have a strength in between stronger covalent bonds and weaker dipole-induced interactions. Unlike the latter, they are directional, with each hydrogen atom pointing towards an electron pair on an oxygen atom.
‘It’s a lot of hydrogen bonding for such a small molecule,’ says Martin Chaplin, chemist and water researcher from London South Bank University in the UK. While water behaves like most liquids at higher temperatures, as it’s cooled the hydrogen bonding starts to play a more important role. ‘They actually force a structured ordered state,’ says Chaplin. As temperature decreases, the additional bonding interactions compensate for the loss of entropy the ordered arrangement causes.
Two liquids
Theories to explain water’s properties have continually cropped up over time. In the 1960s a theory from Russian chemists even suggested that water molecules in capillary tubes could form polymeric chains, known as polywater. This was taken seriously until it became clear that their results were due to experimental contaminants.
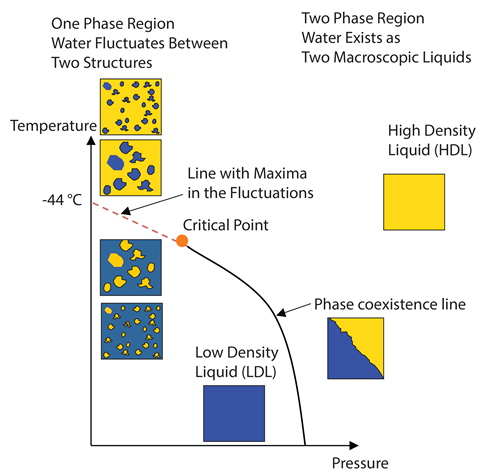
The idea that there may be two different and distinct arrangements of molecules in liquid water goes back over 100 years. German physicist Wilhelm Röntgen , who first discovered x-rays, suggested in 1892 that water was made up of two distinct phases that co-existed in a mixture.
In the 1990s, the two-state model of liquid water returned, based on computer simulations that attempted to replicate the actual behaviour of water. The two forms represent low- and high-density arrangements of the water molecules. The low-density version is a less-ordered ice-like structure, where most molecules are surrounded by four others to generate an open, low-density tetrahedral structure. The higher-density liquid has a higher packing of molecules, and the presence of these additional molecules distorts the hydrogen bonding, producing less-directional and weaker molecular interactions.
In this hypothesis, the proportion of each component changes with temperature, with more low-density water forming as temperature decreases. The result is competition between the expected increase in density with decreasing temperature and the formation of more low-density water. This leads to a density maximum at 4°C, as well as explaining many of water’s other anomalous properties.
Detuning water
A powerful approach to understanding water has been computer modelling. ‘This means coming up with an atomistic model, in which you try to adjust the charges and the electronic distribution in order to reproduce the behaviour of water as accurately as possible,’ says mathematician and modeller John Russo from Sapienza University of Rome in Italy. Russo, together with collaborators at the University of Tokyo in Japan, has created a model that can ‘untune’ the molecular interactions of water molecules to understand the origins of its anomalous properties. ‘I tried to make water less “watery” and try to continuously go from the behaviour of water to the behaviour of a simple liquid,’ he explains.
The key to this process is changing the strength of the hydrogen bonding that leads to the tetrahedral coordination.1 ‘The strength of this interaction can actually be tuned. You can artificially make it more or less tetrahedral.’ By doing this you can tune the structure to vary the macroscopic behaviour of water and, according to Russo, ‘Many anomalies will essentially disappear – you can essentially make water behave like a simple liquid.’ For example, by increasing the ‘tetrahedrality’ – the proportion of water molecules with exactly four nearest neighbours – ice becomes denser than water, and sinks instead of floating, which is what usually happens with normal liquids.
Scientists have created a computer model to make water less ‘watery’
Russo uses a two-state model that treats water as a mixture of the high- and low-density forms, but if these forms do exists, is it possible to isolate them as separate liquids? In 1992, simulations from Eugene Stanley at Boston University in the US suggested that this would occur in supercooled water at very low temperatures.2 Although water freezes at 0°C at atmospheric pressure, without impurities to nucleate crystals, the process is so slow that liquid water can be cooled to about 232K (–41°C). Below this point , water will rapidly freeze, making measurement of the liquid impossible. Water also exists in a glassy state below 150K, but rapidly freezes above this point. ‘Between 235K and 150K is what’s called no man’s land,’ explains Nilsson, where it is not possible to measure the liquid phase at all.
Stanley’s model suggests that supercooled water would actually separate into two distinct phases within this no man’s land, and that a previously unknown critical point exists at such low temperatures. A critical point is the temperature and pressure at which the boundary between two phases disappears; for example, water has a critical point between its liquid and vapour states at 647K and 22.064MPa and the properties of the two phases become indistinguishable. The suggested second critical point in no man’s land would mark the disappearance of the phase separation between two separate liquid phases.
But finding evidence for these two liquid phases isn’t simple. ‘We can’t fill a glass of two kinds of water types, but there have been some experiments which have indirectly seen these sorts of transitions,’ says Russo.
One approach has been looking at low temperature water in its amorphous glassy state. Below 136K two types of glassy water exist – one which has a low density and another one which has a high density. One line of thinking is that these are perhaps equivalent to the low- and high-density water phases that have been proposed and, given the right experimental conditions, it may be possible to detect the separate liquid phases and a transition between them. ‘People are trying this route in order to find evidence for this liquid–liquid transition,’ explains Russo. ‘They start from the glass, they heat it a little bit and try to see if there is a transition to another liquid before it crystallizes [at 150K],’
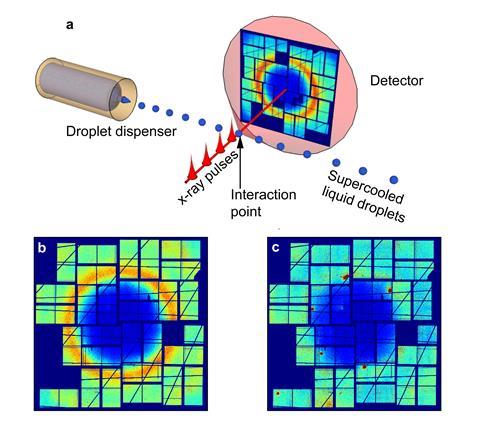
Nilsson has been trying to detect the two liquids using x-ray scattering from ultra-cold water droplets. ‘We have developed techniques to do everything very fast,’ he explains. ‘If you can do [experiments] on the microsecond level, you can push the limit of the no man’s land to lower and lower temperatures.’ His team have cooled 10–20µm water droplets by injecting them into a vacuum, where they evaporate and cool extremely quickly. Using 50–100 femtosecond x-ray laser pulse s, they were able to measure properties related to the structure of any liquid water present in a drop. ‘We’ve been able to go down to 227K, so we have seen what’s been happening inside this no man’s land,’ says Nilsson.3
The Widom line
In 2017 Nilsson’s team published a ground breaking result which they claim provides more evidence for the two-state model of water and helps explain water’s weird behaviour as it is cooled. They found evidence for a Widom line – named after Benjamin Widom, emeritus professor of chemistry at Cornell University in the US. This line projects from a critical point in a phase diagram and, in the case of water, represents a division of the liquid phase into two regions, one where liquid water behaves more like the high-density phase (on the high-temperature, high-pressure side) and the other more like the low-density (on the low-temperature, low-pressure side).
Near this Widom line, the liquid experiences the largest rate of change between the two forms of the liquid and it is possible for one phase to form within the other . The existence of the Widom line and its associated critical point could explain why water fluctuates between a low and high density liquid above the critical point. Even within a single phase, Nilsson’s hypotheses suggests that near the Widom line, very small pockets of the lower density liquid form as temperature decreases, explaining why the density of water decreases from 4°C to its freezing point.
From x-ray scattering experiments of water droplets, Nilsson was able to make electron density fluctuation calculations and identify a maximum value for the compressibility of water at 229K and 0 bar pressure, which would be consistent with the behaviour expected near the Widom line. At this line, the liquid structure undergoes its fastest structural transformation and is therefore at its most compressible.4 Nilsson is continuing to look for the exact position of the critical point. ‘We think it’s probably in the temperature regime near 1kbar [100MPa] at 210K,’ he says.
The skeptics
But not everyone is happy with the hypothesis. ‘There are lots of people who are extremely skeptical about the idea that water is a mixture of two liquids,’ says Alan Soper from the Rutherford Appleton Laboratory in Oxfordshire, UK. ‘Personally I have difficulty visualising how you can have water molecules of two different types.’
Soper says he does not dispute that there may be a second critical point in the no man’s land region, but he says the idea that water might contain patches of two types of water up to ambient conditions implies a phase separation above the critical point. ‘My point is you cannot have phase separation if you’re above a second critical point – it doesn’t make any sense, otherwise it is not a critical point. You can’t have your cake and eat it.’
Soper argues that if patches of two types of water were there, this would be detectable from x-ray scattering experiments. ‘You’d have to have a much higher scattering, and the scattering isn’t there.’ For example, he says, it’s well known from x-ray scattering that mixtures, such as oil and water micelle structures, show huge contrasts between the water region and the oil region, with massive amounts of scattering caused by the density fluctuations between each different region, and this isn’t seen with water.
You cannot have phase separation if you’re above a second critical point
But Nilsson says that he is not suggesting that his patches constitute two phases above the critical point. ‘There are not two phases but are fluctuating regions in the liquid – that is something different from phases.’ He says these may be 50 or 100 molecules in size and constantly interchangeable with the surrounding molecules. He describes the arrangement like guests at a wedding or dinner–dance – in one area, people are moving around and dancing; in another they are sitting at tables – the areas are distinct but people (or molecules) constantly move from one to the other. Nilsson says a simple liquid model just doesn’t explain the anomalous properties water displays.
On the other side, a 2019 x-ray scattering study from a team led by Alexander Föhlisch from Helmholtz-Zentrum Berlin and the University of Potsdam found results consistant with a continuous-distribution model of liquid water at ambient conditions with a level of order that would see each water molecule contributing an average of around 1.75 hydrogen bonds donated and accepted per molecule.5 They concluded therefore that, even if fluctuations can occur near the phase boundary, at ambient conditions a two-state model is unlikely.
What is interesting is we are peering into the region where life exists
Soper thinks that rather than the idea of discrete patches of two liquids, what gives water its unique properties when cooled is the gradual ‘freezing in’ of the orientational order of water molecules in tetrahedral alignment, even when they are still in motion, as would be expected for a liquid. ‘It is this change in the orientational order of water that seems to get overlooked in all the discussions,’ he says and this can explain water’s remarkable behaviour without the need for two distinct states above a critical point.
There is still no consensus among physical chemists as to how water behaves. This may seem a purely esoteric debate, but water’s unusual properties, particularly at very low temperatures, do have implications for other areas of sciences. ‘It affects climate models [at] high pressure, [and] extra-terrestrial water [that] we might find on other planets, or [water in] nano-confinement,’ says Russo. For example, a recent study showed that water at the surface of droplets was much more ordered than bulk water and in a comparable state to supercooled water with strong hydrogen bonding, even at room temp erature.6
Whether a mixture of two liquids or just one, water’s properties are also fundamental to biology. ‘Water is the thing that gives [nucleic acids] their interesting structure and properties, and it’s the same with proteins,’ says Chaplin. Life itself has flourished because ice is less dense than water, allowing organisms to survive underneath floating ice layers. ‘What is interesting is we are peering into the region where life exists when [water’s anomalous behaviour] pops up,’ says Nilsson. According to his two-state model, it is only at temperatures below 50°C that water becomes a mixture of low- and high-density liquid and this is also the temperature region at which life exists. ‘Is this a coincidence, or is there something significant about that?’ asks Nilsson. ‘We don’t [yet] know.’
Rachel Brazil is a science writer based in London, UK
References
1 J Russo, K Akahane and H Tanaka, Proc. Natl Acad. Sci. USA, 2018, 115, E3333 (DOI: 10.1073/pnas.1722339115)
2 P H Poole et al, Nature, 1992, 360, 324 (DOI: 10.1038/360324a0)
3 J Selberg et al, Nature, 2014, 510, 381 (DOI: 10.1038/nature13266)
4 K H Kim et al, Science, 2017, 358, 1589 (DOI: 10.1126/science.aap8269)
5 J Niskanen et al, Proc. Natl Acad. Sci. USA, 2019, 116, 4058 (DOI: 10.1073/pnas.1815701116)
6 N Smolentsev et al, Nat. Commun., 2017, 8, 15548 (DOI: 10.1038/ncomms15548)













8 readers' comments