High-pressure phases of materials can be preserved at ambient pressure by allowing the gaseous material to diffuse into glassy carbon before crushing the carbon into diamond. The process makes some of the most important analytical techniques in modern materials science such as transmission electron microscopy feasible for high-pressure samples, and could even lead to applications for metastable high-pressure phases of matter.
The most common tool in high pressure physics is the diamond anvil cell. Though refined hugely since its invention in the 1950s, it remains essentially a giant set of nutcrackers, in which a sample is mounted in a metal gasket between two wedge-shaped diamonds that are then squeezed together. Diamond anvil cells have applied pressures exceeding one terapascal (10 million times atmospheric pressure) and generated novel superconductors, helium compounds and multiple claims of metallic hydrogen. One problem, however, is that analysis of the resulting materials must be performed through the diamonds – which rules out many state-of-the-art techniques.
One simple solution would be to put the material inside another material that undergoes a phase change under pressure. Then, when the external pressure is removed, the material under study could remain trapped inside. Nature itself does this: diamonds often contain inclusions of pressurised material, for example, and though jewellers consider them flaws, they can contain information about the conditions deep inside the Earth where they formed. Scientists have therefore tried to create artificial diamonds and other materials with inclusions. This has hitherto proved impossible, however: ‘If you use a porous material like graphite filled with gases or liquids and you try to synthesise diamonds you should expect the pores will close. But because the pores are all elongated and connected to one another all the gas or liquid you trap will escape,’ explains Vitali Prakapenka of the University of Chicago in the US.
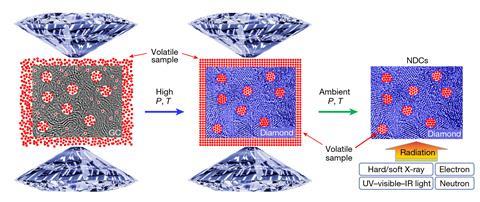
Wendy Mao of Stanford University and colleagues in the US and China therefore used glassy carbon – an amorphous material comprising numerous broken, fullerene-like pieces. ‘It’s not very porous under ambient conditions, but at high pressure you can start squeezing gases into the pores,’ explains Mao. The researchers therefore used a diamond and cell to compress samples of glassy carbon in both argon and neon atmospheres to up to 50GPa before using an infrared laser to heat the materials, inducing transitions to diamond. When they released the pressure, they found the crystalline forms of the noble gases were preserved as inclusions.
They analysed these materials using techniques such as soft x-ray spectroscopy and transmission electron microscopy that would previously have been impossible. Mao explains that the group’s findings about the materials are consistent with previous results: ‘We really wanted to demonstrate the strategy,’ she says. ‘If we had found something truly weird in the quenched material then people would have said, “Well, you didn’t quench what happened at high pressure”.’ The researchers now hope that their ability to use previously unfeasible techniques may provide genuinely new insights into materials, although she declines to say what the group’s next targets are.
‘It’s a very nice finding,’ says Prakapenka, who has worked on several similar projects. ‘You can synthesise many unique materials at high pressure, but usually when you quench they convert back.’ However, materials such as hydrides that have shown superconductivity near room temperature at high pressure could potentially retain it at ambient pressure in systems such as these.
References
Z Zeng et al, Nature, 2022, 608, 513 (DOI: 10.1038/s41586-022-04955-z)












No comments yet