Drops of hexadecane adopt geometric shapes more appropriate to crystals
What shape is a droplet? Surface tension usually decides the answer – minimising the surface area, since interfaces cost energy, it pulls a raindrop into a sphere and a floating oil droplet on water into a circular lens. But Stoyan Smoukov from the University of Cambridge, UK, and colleagues have found that sometimes the question isn’t so simple. They have shown that liquid drops of oily hexadecane, coated with a surfactant and floating on water, can adopt geometric shapes seemingly more appropriate to crystals, with hexagonal, rhombus and triangular profiles, as well as more elaborate shapes in which long ‘tentacles’ protrude from the corners of these polygons.
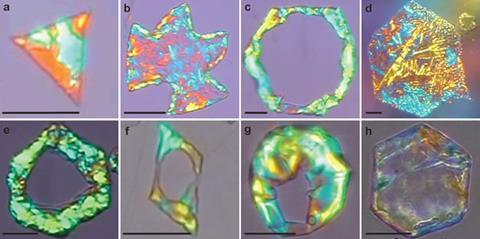
The weird shapes were completely unexpected, appearing as the researchers studied regular emulsions (mixtures of immiscible fluids stabilised by surfactants). ‘My colleagues had noticed a few strange shapes while freezing droplets, and we decided to explore these further,’ says Smoukov. Both the regularity of the shapes and the potential to control them, he adds, were ‘beyond surprising’, according to team member Slavka Tcholakova of Sofia University in Bulgaria. ‘The simplicity of this shape-forming system make us suspect it may be used somewhere in nature,’ she adds.
The researchers find that droplets of all alkanes with chains 14–20 carbon atoms long exhibit the same behaviour, in combination with various surfactant types. The only requirement for achieving the polygonal droplets is that the surfactant that coats the oil–water interface should have chains at least as long as those of the alkane oil itself. With 1.5 weight percent of surfactant dispersed in the water, Smoukov and colleagues see a series of shape changes as initially circular droplets of hexadecane, several tens of micrometres across, are slowly supercooled below the alkane’s bulk freezing point (18°C), but while the drops still remain liquid. First they adopt a three-dimensional octahedral shape, followed by hexagonal platelets which then become either rhomboidal or triangular plates. Then the corners sprout thin arms, which grow into long tendrils. Eventually the droplets freeze throughout, whereupon their appearance in the microscope abruptly changes from transparent to brightly coloured owing to light-interference effects.
Crowd theory
What causes this behaviour? The researchers think that the surfactants packed side by side on the surface of the droplets freeze first into an orderly monolayer, which then induces some of the alkane too to form a kind of skin at the surface. This layer is a so-called rotator phase, in which the molecules are regularly packed but still free to rotate, like people turning around in a dense crowd. This rather thick and stiff skin gives the flat droplets semi-cylindrical edges that resist bending along the cylindrical axis.
But they can’t be wholly straight, because they have to form a closed perimeter around the alkane drop; so all the curvature is concentrated into sharp kinks creating a kind of geometric frame at the droplet periphery. ‘We think the corners of the polygons are defects in the rotator phase,’ says team member Nikolai Denkov in Sofia. Initially six evenly spaced corners produce hexagons. But the defects can merge into pairs, producing either rhombuses or triangles. The tendrils, meanwhile, are pure cylinders of the surfactant-alkane rotator-phase skin.
The researchers say their results offer a scaleable, efficient way to produce microparticles (and perhaps still smaller ones) with ‘shape on demand’. They are looking into ways of fixing the shapes by polymerising the skin. It’s not clear yet what they might be used for. ‘There is some evidence of different shapes being taken up differently in different organs,’ says Smoukov, ‘so there may be some potential for selective drug delivery.’
Sam Safran of the Weizmann Institute of Science in Israel says that the results are interesting, and could shed light on other problems in crystal growth, such as the way alkanes can form a frozen surface monolayer above their bulk freezing temperature. He also wonders if the ‘internal’ solid–liquid interface between the crystalline rotator phase and the liquid inside the droplet will have a role in the energy balance that seems to determine the shapes. However, he points out that, as the shapes depend on the cooling rate, they must be a non-equilibrium effect. So there’s plenty still to be understood about the system.
References
N Denkov et al, Nature, 2015, 528, 392 (DOI: 10.1038/nature16189)











No comments yet