Mechanical activation converts a strong new polymer into an alternative chemical form that more easily degrades into simple monomer units. The idea could offer a way to regenerate valuable chemical feedstocks from discarded plastics.
Plastics are ubiquitous in modern life. Their chemical inertness and durability make them ideally suited to thousands of diverse applications, but limit the ease with which they can be recycled. The strong carbon–carbon bonds of the polymer backbone are challenging to break, and current recycling methods generally reuse the polymer chain rather than recovering the chemical feedstocks that are locked within it. Degradable plastics incorporate other functional groups into the polymer backbone but this tends to reduce their stability, making them unsuitable for most commercial uses.
Now, a team led by Junpeng Wang at the University of Akron, US, has fused these seemingly contradictory properties to design a material able to switch from a stable plastic into a readily degradable polymer. The polymer design incorporates two distinct molecular forms: a ‘locked’ form with a robust carbon backbone, and an ‘unlocked’ form containing simpler and more reactive repeating units. Mechanical activation by sonication or ball milling converts the durable locked plastic into its degradable form, allowing recovery of the valuable chemical feedstocks.
‘We wanted to break the paradox between degradability and stability,’ explains Wang. ‘By leveraging a strategy called locked degradation, we created a polymer which is stable during storage and use, and when recycling is needed, its degradability can be unlocked.’
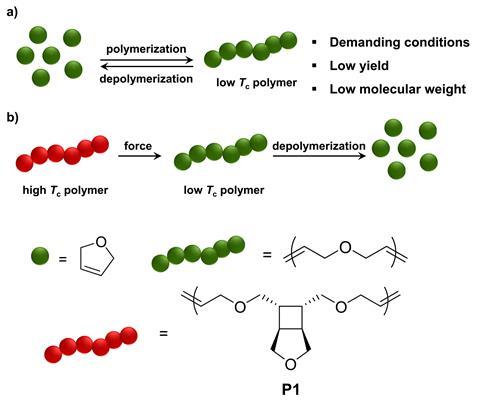
The team carefully selected a strained ring system (cyclobutane-fused THF) as the repeating unit for the locked polymer, producing a tough plastic similar to high-density polyethylene. Under mechanical force, the strained rings of each monomer unit open to produce three simpler repeating units, all containing a carbon–carbon double bond. Once unlocked these reactive alkene handles enable commercial catalysts to fully depolymerise the chain. ‘The unlocking step brings degradable functional groups to the polymer backbone,’ says Wang. ‘The mechanochemical step turns the cyclobutane ring into carbon–carbon double bonds, which can then undergo metathesis-based degradation in the presence of Grubbs’ ruthenium catalysts.’
‘The work expands on a creative concept, and provides a nice example of using mechanochemistry to access polymers that are otherwise quite difficult to make,’ says Stephen Craig, a polymer chemist from Duke University, US. ‘Longer term challenges include improving the practical circularity of the polymerisation/depolymerisation [and] identifying conditions for higher yield mechanochemical conversion on larger scales.’
The team are currently investigating producing and recycling the polymer at a larger scale and lower cost, but Wang hopes that the work will have a wider impact. ‘We can now enable effective recycling without compromising the stability of polymers,’ he says. ‘Ultimately, we’re aiming to apply the concept of locked degradability to existing commercial polymers too.’
References
T-G Hsu et al, Nat. Commun., 2023, DOI: 10.1038/s41467-023-35925-2

















No comments yet