Researchers in the US have synthesised the first macrocyclic N-heterocylic carbene (NHC) complexes to incorporate elements from the f block and investigated their unusual electronic structures.
Despite the widespread use of NHC ligands in organometallic complexes, examples involving the actinides are scarce because an electronic mismatch between them hinders ligation of neutral NHCs. Polly Arnold, now at the University of California, Berkeley, and her group developed synthetic strategies to avoid these issues by using chelating ligands with anionic groups, and others have used bis- or tris-NHC borate ligands with a delocalised negative charge across a polydentate backbone.
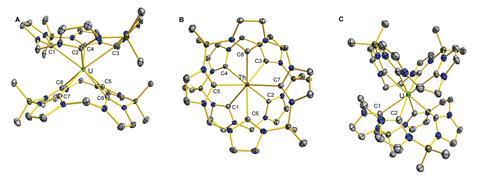
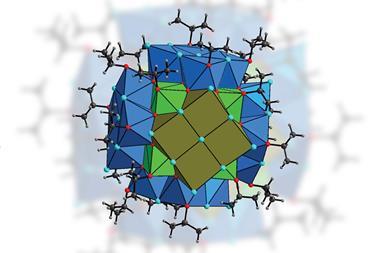
David Jenkins, from the University of Tennessee, and his team recently developed dianionic tetra-NHC complexes for catalysis and transition metal group transfer reactions. Now, a team surrounding both Arnold and Jenkins has tested whether macrocyclic NHC ligands could better stabilise larger actinide cations. Combining expertise from both groups saw them create octa-NHC actinide complexes with interesting electronic structures due to the strong σ-donor ability of the NHC ligand.
Synthesising the octa-NHC actinide complexes was challenging; analogous strategies for transition metal systems using strong bases such as n-butyllithium did not translate to the actinide variants. Instead, they added sodium bis(trimethylsilyl)amide to deprotonate the ligand, followed by the actinide species and heat.
They created three complexes with different geometries depending on the macrocycle used. Complexes containing square-antiprismatic uranium or thorium sandwiched between two 16-atom macrocycles, were stable in air, and a complex containing square prismatic uranium sandwiched between two 18-atom macrocycles, was reactive and unstable.
The researchers investigated the complexes using density functional theory and compared their results to the original actinide sandwiches, uranocene and thorocene. They noted that the orbital picture for the new uranium complexes and uranocene was quite similar. However, the HOMO–LUMO gap for the new thorium complex was larger than thorocene’s, indicating that the σ-donor NHC ligand had a stronger ligand field than thorocene’s π-aromatic cyclooctatetraene ligand.








No comments yet