Scientists have designed and made the first stable chiral compound containing a positively charged oxygen species as the only centre of asymmetry. To accomplish this, they studied oxonium ions, in which an oxygen atom is connected to three other atoms. ‘Such compounds are typically reactive and short-lived, while the oxygen atoms also tend to quickly flip between mirror-image forms,’ says Robert Paton from Colorado State University and the NSF Center for Computer Assisted Synthesis, US, who led the study together with Martin Smith and Jonathan Burton at the University of Oxford in the UK. Paton explains that because of these properties, it hadn’t been possible to isolate oxonium ions as a single enantiomer before. ‘Using a combination of synthetic, analytical and computational studies, we have been able to establish new design rules how to capture a stable chiral oxygen atom,’ he adds.
Nick Tomkinson at the University of Strathclyde, UK, who wasn’t involved in the study, says that this is an exciting method for introducing chirality into a molecule. ‘Chiral molecules represent the foundation of asymmetric catalysis, which is used in the preparation of medicines, food additives and fragrances. This work has the potential to open up new unexplored areas of asymmetric catalysis.’
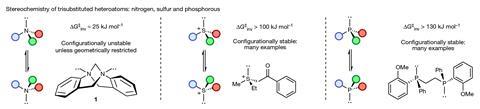
While the chirality of carbon atoms has been widely explored and there has also been a lot of work on the chirality of trivalent nitrogen, reports on the inversion rate of oxonium ions are extremely scarce, points out Smith. ‘We found that triaryl oxonium ions – which were first reported in the 1950s – were isolable, stable and crystalline solids, so we felt that they could be a good candidate to synthesise and analyse,’ he says. ‘We rationalised that embedding the trivalent oxygen atom in a relatively inflexible ring system would slow down the inversion process, and so our primary task was to work out the ring size and substitution pattern that would be most effective.’
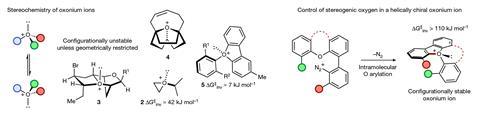
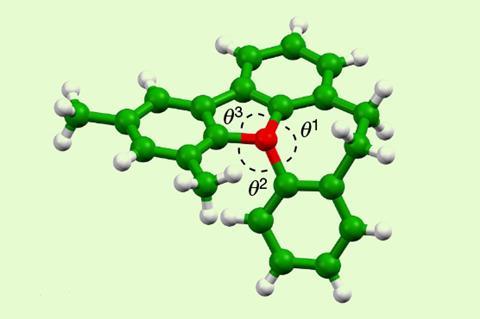
The team evaluated potential candidates theoretically. ‘With the aid of quantum mechanical calculations, the final molecule we arrived at contains a central positively-charged oxygen atom directly attached to three aromatic substituents that are tethered together by five-atom and seven-atom rings,’ explains Paton. He clarifies that the additional ring systems give the molecule a helical-like three-dimensional structure that slows racemisation.
Tomkinson compares the oxonium ion with an umbrella that can turn inside out in a strong wind. ‘Through incorporation of the oxygen atom within the helix, the oxonium ion is prevented from losing its stereogenicity,’ he says.
The researchers examined the inversion rates using NMR spectroscopy and liquid chromatography and studied the configuration of the compounds by x-ray diffraction. ‘This showed that the seven-ring-containing oxonium ion has a half-life to inversion of more than a month at room temperature and, as predicted, is indefinitely stable,’ notes Smith.
Mark Mascal from the University of California, Davis in the US mentions that the new concept is an important milestone in chemistry. ‘This work is a very nice example of fundamental science, which sets out to test the limits of the bonding and structure of molecules,’ he says.
Stephan Rigol at Rice University in the US adds that the results ‘can broaden our knowledge of oxonium ions and potentially open a novel entry to their exploitation in future organic synthesis endeavours’.
Because of the importance of chirality in catalysis, medicine and materials, Paton believes that it will be exciting to explore the properties of chiral oxygen-atom-containing compounds in future studies. ‘The discovery of this fundamentally new example of molecular chirality demonstrates our ability as chemists to design and synthesise new matter based on a computational blueprint.’
References
O Smith et al, Nature, 2022, DOI: 10.1038/s41586-023-05719-z











No comments yet