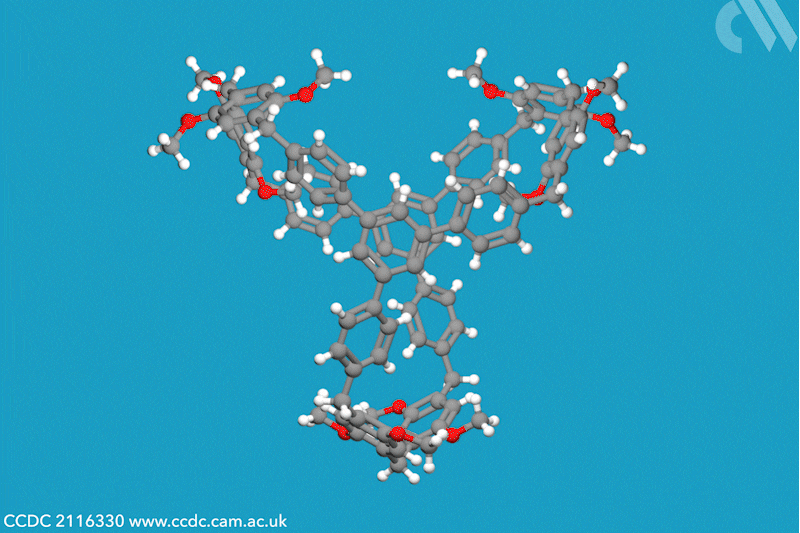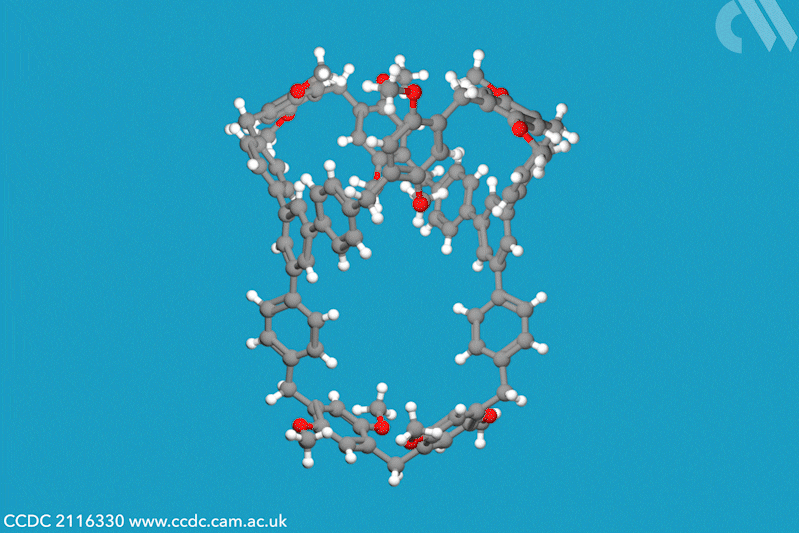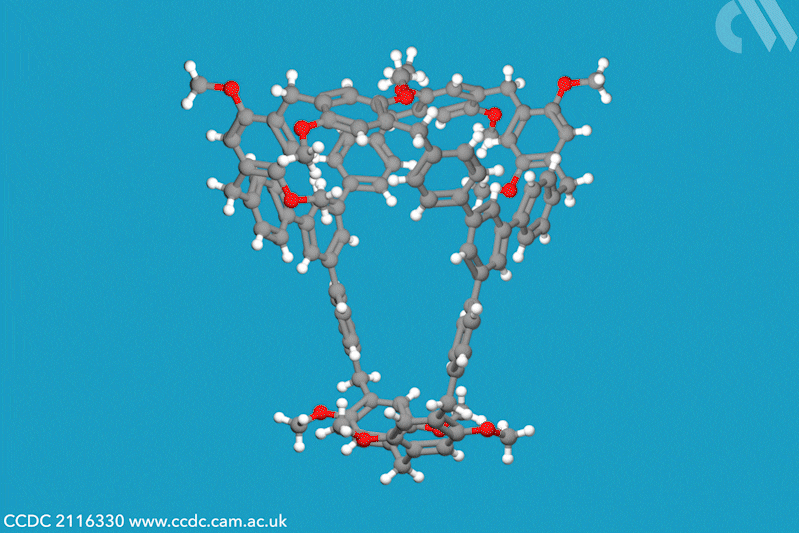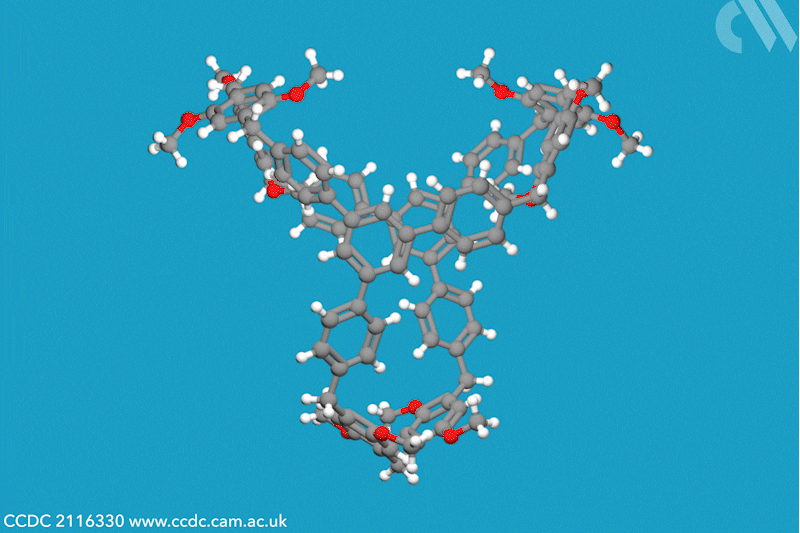
A new family of molecular cages has been synthesised using classical covalent chemistry, overcoming the limitations of both supramolecular and self-assembled organic frameworks. These cages, dubbed cagearenes, are able to separate hydrocarbon mixtures through a process called programmed release, which has widespread applications in both drug delivery and chemical production.
Programmed release depends on the interactions of a host framework with a smaller guest molecule. Supramolecular structures require the guest molecule to sit within the host cavity and typically exhibit poor separational selectivity due to the uniformity of interactions experienced by all guest molecules. Self-assembled organic frameworks open up the possibility of creating different interactions with different guests but suffer from poor structural and chemical stability, arising from the inherent reversibility of their bond formation.
Researchers in China have now constructed two organic cages from irreversible covalent bonds, using the Friedel–Crafts reaction to create a sprawling aromatic precursor. At high temperatures, two molecules of this precursor condense with three molecules of paraformaldehyde to form the hollow cagearenes.
Thermogravimetric analysis experiments revealed the cagearenes’ differing affinity for the similar hydrocarbons benzene and cyclohexane, indicating that these organic frameworks could be used for purification. The team proposes that this programmed release is possible due to the different interactions of each guest molecule with the cagearene, a hypothesis supported by x-ray crystal analysis. Benzene is weakly adsorbed to the surface of the cage and is released upon heating to 70°C. Conversely, cyclohexane forms a very stable host–guest complex within the cavity and requires a much higher temperature for release. The team is now working to create functionalised cages for a wider range of applications.








No comments yet