Microfluidic device shows that antiperspirants control sweating by forming pore-plugging aggregates with perspiration proteins
Researchers in France have made a microfluidic device that mimics sweat pores, showing that perspiration proteins are essential to the way deodorants work.
Antiperspirants control sweating through the interaction of aluminium salts with sweat. A number of theories have been put forward for how these products work; the most widely accepted one being that the salts diffuse into the sweat duct and hydrolyse in contact with sweat, forming an aluminium hydroxide plug that blocks sweat flow.
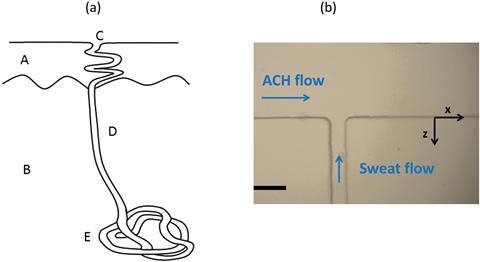
The truth maybe a little more complicated, however, as Bernard Cabane from L’Oreal, Jean-Baptiste Galey from ESPCI ParisTech and colleagues have discovered. They have built a microfluidic T-junction that mimics sweat ducts, and allows them to study the interactions between sweat and aluminium chlorohydrate (ACH) in real time. The device consists of a surface channel filled with ACH, and an orthogonal channel into which they pump sweat. Using time and spatially resolved small-angle x-ray scattering, they studied the structure and composition of the plugs formed, and how they block the sweat duct.
The results show that ACH aggregates with proteins in the sweat, forming a membrane that extends across the duct. It then collects further proteins and grows to form a plug that blocks the duct entirely. ‘Contrary to previously published works, we demonstrate that sweat proteins are necessary for pore plugging, in spite of their very low concentration, because they are constantly brought by the flow,’ says Cabane.
‘This team has developed an elegant microfluidics device to mimic the sweat gland,’ comments Robert Prud’homme, an expert in molecular level interactions at Princeton University in the US. ‘They might have stopped with just the visual observations of the formation of plugs, but they have upped the ante and conducted beautiful small angle scattering experiments to enable quantitative statements about the protein aggregation driven by aluminium ion binding.’
Galey thinks these findings could help develop improved personal care products in the future. ‘We hope these results will help to find new antiperspirants with improved efficacy based on the size and charge of reactive species found in the present work, and on their ability to react with sweat proteins,’ he explains.
References
This article is free to access until 29 June
A Bretagne et al, Soft Matter, 2017, DOI: 10.1039/C6SM02510B












No comments yet