Scientists working in Israel and Spain have used two-dimensional NMR to monitor a reaction in real time
Chemists have created an ultrafast NMR technique that can ’watch’ how chemical reactions occur in real time. They’ve used the technique to follow the formation of pyrimidines from carbonyls and even identified new intermediates never before thought to be part of the reaction.
Multidimensional NMR is rich with information. The details of atomic connectivity and proximity it provides make it an essential analytical tool. But the information comes at a price: long experiment times and, often, the need for costly and laborious isotopic labelling of samples. These drawbacks have limited the use of multidimensional NMR in dynamic investigations.
To monitor real-time chemical processes, you need to acquire spectra on the same timescale as the processes that are being observed. This demands faster and faster NMR techniques - and the ultrafast NMR technique, developed by Lucio Frydman and his team at the Weizmann Institute of Science in Israel, is the fastest around. In 2010 the team used this technique to observe the folding of RNA in real-time1 but now, for the first time, the technique has been applied to organic synthesis and with natural abundance samples.2
In conventional two-dimensional NMR, the experiment is repeated many times with each additional experiment increasing the resolution of the second dimension, but also increasing the experiment time, often to hours. In ultrafast NMR, these experiments are carried out simultaneously along the length of the sample to create a complete two-dimensional spectrum in milliseconds. ’It’s quite spectacular when you think about it - you can acquire thousands of two-dimensional spectra in an hour,’ says Dusan Uhr?n, a lecturer in NMR at the University of Edinburgh, UK. ’You could follow a reaction with a series of one-dimensional experiments, but [ultrafast two-dimensional NMR] increases the information content so a chemist can interpret the mechanism.’
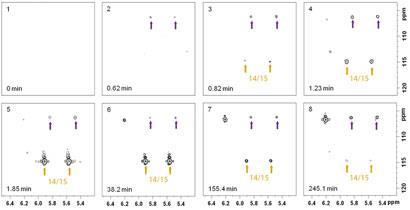
Frydman’s team investigated the synthesis of pyrimidines via the reaction of carbonyls - a well known reaction but one whose mechanism and kinetics are still largely unknown. ’We wanted to exploit the information available in heteronuclear two-dimensional correlations in order to identify the nature of the intermediates,’ says Frydman. By rapidly acquiring a series of two-dimensional spectra, an NMR ’movie’ of the reaction was created, much like brain imaging in functional MRI. By observing peaks appearing and disappearing as the reaction progressed, the team could then identify and assign the reaction intermediates. ’This is extremely powerful,’ says Eriks Kupce, a fellow at Agilent Technologies. ’Allowing the user to "participate" in the reaction ... takes analytical chemistry to a whole new level.’
Intriguingly, not only were previously proposed intermediates confirmed, the team also identified some new intermediates, leading to a novel mechanism being proposed. ’This is an exciting development,’ says Chris Moody, a professor of synthetic chemistry at the University of Nottingham, UK. ’In the synthesis of heterocyclic rings, it is rare that the mechanisms have been established beyond doubt. This research allows the intermediates to be clearly identified, thereby revealing the mechanism.’
Patrick Giraudeau, a researcher in NMR at the University of Nantes, says that the time is ripe for the widespread application of this technique. ’This is a great demonstration of what can be done with ultrafast NMR,’ he says. ’I really think chemists need to be aware of these developments. If we communicate better, if we advertise these techniques, they will become routine.’
Frydman, meanwhile, is confident the technique can become faster and more sensitive. ’Eventually we will get the speed, with all the sensitivity, the dynamic information and atomic site resolution that only multidimensional NMR can afford,’ he says. ’If there is a field that has always found unexpected solutions to its problems, that field is NMR.’












No comments yet