The products of thermally exfoliating graphite oxide to make graphene are much more complex than previously thought, new research shows. The volatile compounds formed vary with reaction conditions, and may influence the graphene’s structure.
The most common way to prepare graphene is by thermally reducing – or ‘exfoliating’ – graphite oxide. But the graphene produced often contains defects and lacks the perfect honeycomb structure. One explanation is that these defects may be the result of organic by-products forming and escaping as gases during the reaction.
‘It has been commonly believed that the only gaseous products of graphite oxide exfoliation are water, carbon monoxide and carbon dioxide,’ explains Martin Pumera from Nantang Technological University, Singapore, who led the work. ‘But this belief was based on only a few studies which never looked for anything more complex.’
Pumera and his colleagues exfoliated graphite oxide in an autoclave at 500°C for two hours, then analysed the gases produced using gas chromatography-mass spectrometry (GC-MS). They detected a large number of volatiles, including many polycyclic aromatic molecules, and several containing sulfur and nitrogen from contaminants in the graphite oxide starting material.
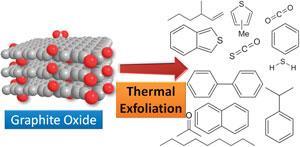
Understanding these by-products is crucial, as they can affect the structure of the resultant graphene, which influences its future use, says Pumera. He suggests that measuring the volatiles produced during exfoliation could help determine the nature of defects.
Pance Naumov, a materials chemist at New York University Abu Dhabi, United Arab Emirates, agrees that the findings have implications for routine exfoliation of graphite oxide. ‘These observations should be taken into account in future plans for large scale production of graphene using this methodology,’ he says.












No comments yet