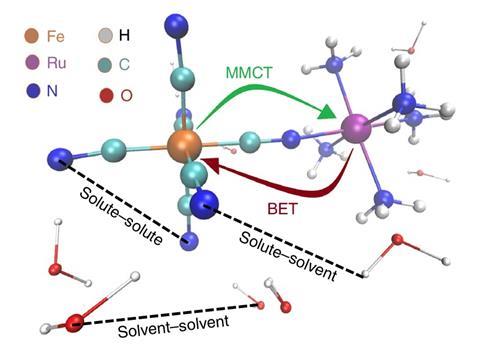
The rapid rearrangement of water molecules around a bimetallic complex following a charge-transfer process has been monitored in real time and with atomic resolution by combining x-ray techniques with molecular dynamics simulations. The new results shed light on how charge flow within a molecule is coupled to the molecule’s environment, explains Elisa Biasin from the SLAC National Accelerator Laboratory in the US. ‘This systematic and quantitative understanding of the molecular interactions will help us unravel complex mechanisms in natural and artificial energy-conversion processes,’ she says.
Electron-transfer reactions are important in energy harvesting. Although the solvent is known to play a crucial role in such processes, the molecular details of solute–solvent interactions remain largely unexplored. ‘This has been a huge challenge due to the very short timescales involved,’ comments Kristoffer Haldrup at the Technical University of Denmark, who did not participate in the study. ‘But with work such as this we can now, for the first time, directly see what nature is doing and compare it to our models.’
Carlos Baiz, a researcher at the University of Texas at Austin, US, agrees that investigating the motion of molecules on the femtosecond timescale is one of the frontiers of ultrafast science. ‘The main challenge stems from being able to isolate the signals from the small subset of molecules around the solute from the rest of the molecules in the liquid,’ he says.
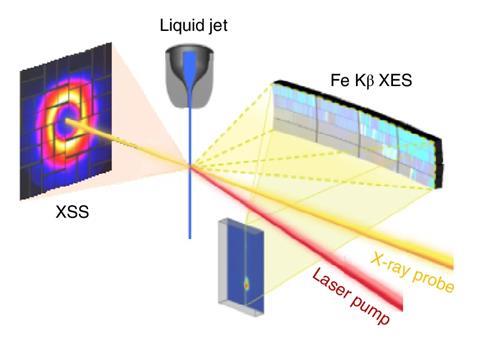
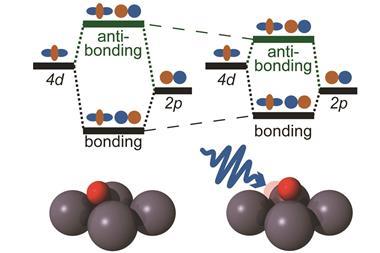
Using a combination of x-ray scattering, x-ray emission spectroscopy and molecular dynamics simulations, Biasin and colleagues have now been able to study the rapid motion of solvent water molecules during light-driven charge transfer in an iron–ruthenium complex. The team used the Linac Coherent Light Source x-ray free electron laser at SLAC to conduct the experiments, combined with advanced simulations on supercomputers to analyse and interpret the obtained data. They found that the charge-transfer excited state had a lifetime of 62 femtoseconds (10-15 s)and that reorganisation of the first solvation shell affected the back electron transfer.
‘We first optically excited the molecule to initiate electron transfer between the iron and ruthenium atoms and then used x-ray pulses as an “ultrafast camera” to record the resulting motions,’ explains Biasin. ‘The x-rays that get scattered contain information about the positions of the atoms comprising the bimetallic complex and the water molecules around it with exquisite resolution,’ she says. Biasin adds that the simulations were crucial for identifying the subtle translational motions of the water molecules surrounding the complex, which she says arise from changes in hydrogen bonding.
‘The authors truly broke new ground by pushing the time resolution down into the sub-100 femtosecond regime, an achievement made possible by combining structural investigations with x-ray spectroscopy,’ points out Haldrup. ‘This allowed them to “clock” the reaction with very high accuracy.’
Adam Kirrander of the University of Edinburgh, UK, who recently used x-rays to observe the initial step of a light-driven reaction, says that the combination of x-ray scattering and x-ray spectroscopy is a powerful tool for detailed mechanistic studies. ‘This experiment demonstrates, beautifully, the atomistic level detail that can be achieved by combining complementary techniques and the important role of simulations to interpret the experimental data. I am convinced that we will see more and more of such multimodal measurements in coming years, as these allow for unprecedented insights into complex systems.’
References
E Biasin et al,Nat. Chem., 2020, DOI: 10.1038/s41557-020-00629-3









No comments yet