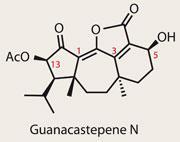
Guanacastepene N
Guanacastepene N
After successfully working out a neat piece of synthetic methodology, sometimes finding an appropriate natural product to showcase it can be tricky. Multiple literature searches show only fragments of similarity, and the researchers resort to a lab-based crowbar. However, in other cases, the choice is more obvious - such as that encountered by Erick Carriera and his team at the Swiss Federal Institute of Technology (ETH) in Zurich. Their work on cycloadditions of exotic and reactive species1 naturally lends itself to ’medium ring’ based targets, of which the guanacastepenes are notable examples.2
Discovered in fungi growing on trees in the Guanacaste conservation area in Costa Rica, several syntheses of this family have appeared in the decade since their isolation.3 Their potent antibiotic activity has secured interest (and funding), but their concomitant toxicity has hindered their medical application. However, this kind of problem is exactly what gets the more medicinally minded organic chemist excited, as there may be analogues that posses the desirable activity without the toxicity. What’s needed, then, is a modular synthesis, where adaptation of the key fragments would result in efficient synthesis of analogues - an art in which the Swiss team has considerable experience.
Ringing the changes
The team’s approach starts with a neat literature synthesis of a cyclopentenone in two steps. A few steps and a second stereocentre later, an intramolecular aldol condensation closes the second ring to give a mixture of two epimeric alcohol products. The lack of stereocontrol in this step was of no consequence, as the alcohol was oxidised to a ketone in preparation for the team’s key cycloaddition chemistry.
A brief examination of the reagents for the cycloaddition proves that something quite interesting is going on. We’re looking at a hypervalent iodine reagent. But unlike earlier in the synthesis, where hypervalent iodine acted as an oxidant, its role in this reagent is to generate a rather exotic intermediate - cyclohexyne (figure 1). The reactivity of this species can be easily proven by grabbing a molecular modelling kit and attempting its construction - which can’t be done without many bent and broken pieces of plastic tubing.
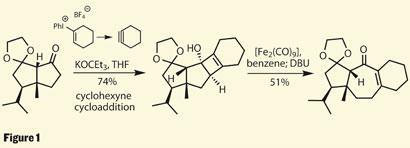
This instability encourages a rather unusual [2+2] cycloaddition with the enolate of the ketone fragment, building yet another strained ring, this time a cyclobutene (you may try with the molecular modelling kit again, but I suggest wearing safety specs). Luckily, this species was stable enough to progress to the next step, a rearrangement to the less precarious world of medium rings, using a rather unusual iron carbonyl complex. With a little base, the rearrangement was complete, neatly forming the 5,7,6-ring system peculiar to the guanacastepenes.
Take the strain...
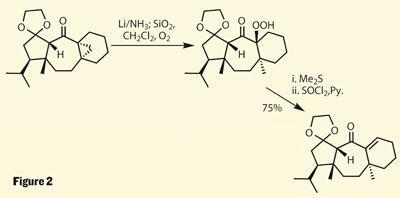
The group then decided to add yet another strained ring, this time the less exotic cyclopropane. Yet again, the strain in the molecule allows for some rather unusual chemistry, as a mixture of ’electron soup’ (lithium in ammonia) and oxygen leads to fragmentation of the three membered-ring to provide an additional methyl group at the ring junction, as well as re-forming the double bond in a more useful position (figure 2).
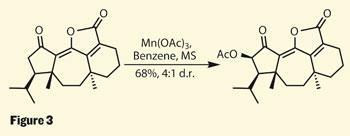
One of the later reactions in this synthesis of guanacastepene N, concerns a reagent close to my heart - manganese triacetate. Brown, cheap and smelling quite strongly of acetic acid one might not expect much discussion of what appears to be the least interesting reagent in this paper. But not only does it fulfil a rather handy late-stage oxidation (figure 3), it was also the subject of this columnist’s PhD thesis.4 However, I never thought of doing that with it!
Paul Docherty is a science writer and blogger based in Reading, UK
References
J. Am. Chem. Soc., 2000, 122, 2116
4 L H Powell et al, Chem. Commun., 2008, 2559












No comments yet