Just because electrolytes are effectively inert, doesn’t mean they can’t be sophisticated
Battery developers may have just been honoured by the 2019 Nobel prize in chemistry, but that doesn’t mean the technology is anywhere close to being the finished product. Whereas a couple of years ago we often brought you news stories about new electrode materials that promised to enhance batteries, I’m noticing a lot more research into alternative electrolytes.
The ideal battery electrolyte transports ions like a liquid but resists morphological and hydrodynamic instability like a solid. Being cheap and simple to make are also a given. That said, electrolyte research is attracting an array of sophisticated materials, such as superhalogens, molten salts and self-healing polymers.
Much of John Goodenough’s battery research has concerned cathode materials. Yet, the latest research from our oldest ever Nobel laureate, published just seven days before his prize was announced, concerns a polymeric network capable of trapping liquid electrolytes.1 Immobilising the liquid electrolyte during cycling significantly reduces side reactions during cell operation but still transports lithium ions quickly between the electrodes. Goodenough and his University of Texas at Austin colleagues say the benefits of this system make it appropriate for use with metallic lithium or sodium anodes and therefore large-scale applications.
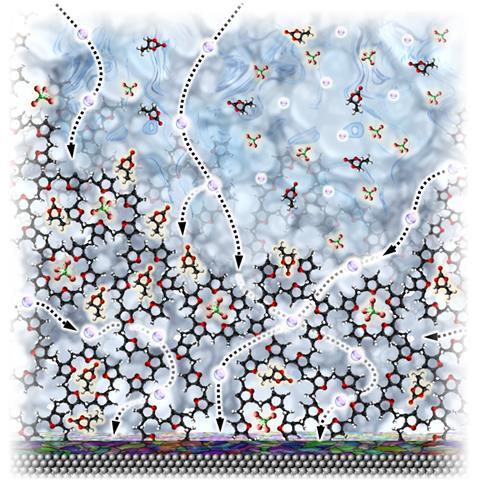
We also featured some new electrolyte research at the beginning of October, this time for calcium batteries. Calcium’s low electronegativity, double valence, abundance and low cost mean it has huge potential as a component material in high voltage grid-scale batteries. Eliminating the logjam created by a lack of compatible electrolyte means researchers can crack on with capitalising on those properties.
Enhanced safety is a typical objective for much electrolyte research and tests to measure electrolyte flash points and self-extinguishing times are increasingly prominent in such studies. Demands for ultrafast rechargeable devices means we are seeing battery systems with more oxidising cathodes and more reducing anodes. Researchers are tailoring the electrochemical windows of electrolytes to support those different electrode chemistries and maximise safety.
The right electrolytes are central to building rechargeable batteries that can deliver safe, low-cost and high-energy storage. Without such batteries, society will fail to manage the intermittent nature of renewable energy. Lithium-ion batteries, and their cousins, have undoubtedly enhanced our lives but society is now relying on them to save our planet.
References
1 H Khani, S Kalami and J B Goodenough, Sustainable Energy Fuels, 2019, DOI: 10.1039/c9se00690g










No comments yet