From their origins in the 1970s oil crisis to powering 5 billion phones around the world, Katrina Krämer traces the story of how lithium-ion batteries earned John Goodenough, Stanley Whittingham and Akira Yoshino the 2019 Nobel prize
On Wednesday 9th of October, Maria Helena Braga received a message from her son. It said simply ‘He won!’
Braga, a solid-state physicist at the University of Porto in Portugal, immediately knew what it meant: her close collaborator John Goodenough had won the chemistry Nobel prize, sharing it with Stanley Whittingham and Akira Yoshino for developing the lithium-ion battery.
At the time the prize was announced, Goodenough and Braga were in London, celebrating Goodenough’s award of the Copley Medal, the world’s oldest scientific prize. Although his invention helped launch the mobile revolution, Goodenough doesn’t own a mobile phone and the Nobel committee was unable to reach him.
‘Somebody with a cell phone managed to get the information on the phone this morning while I was brushing my teeth,’ he laughed in an interview with Nature on the day of the announcement. ‘Of course I was very pleased. I suppose I was surprised, but at my age, it doesn’t really make much difference.’ At 97 years of age, Goodenough is the world’s oldest Nobel laureate.
‘It was really exciting and long overdue,’ says Yang Shao-Horn, a battery researcher at the Massachusetts Institute of Technology in the US. ‘I think many of us in the community have been hoping each year that this contribution would be recognised.’
For years, lithium-ion batteries had been hailed as the next Nobel-winning invention, with the trio’s names regularly popping up in polls and predictions come Nobel season. ‘It was really overwhelming,’ says Braga. ‘We were thinking it would come, but who knows? It was really nice that [Goodenough] lived to see the time when the announcement finally came.’
Having access to cheap, safe and powerful batteries has impacted society profoundly. Today, over 5 billion people – 66% of the world’s population – own mobile devices. Energy storage in the form of batteries is driving development in countries lacking electric grid coverage. In the Democratic Republic of Congo, solar cells combined with batteries are set to supply power to 2.5 million of its off-grid citizens.
We were thinking the prize would come, but who knows?
Batteries are also starting to transform the transport sector. A decade ago, there were almost no electric cars, while now around half of all lithium-ion battery capacity sold is used in the automotive industry.1 Over time, increasingly powerful batteries charged by sustainable electricity might help wean humanity off their transport-driven addiction to fossil fuels.
For their work kick starting the mobile revolution, Goodenough from the University of Texas at Austin, US, Whittingham from Binghamton University, US, and Yoshino at the chemical company Asahi Kasei and Meijo University, Japan, are now sharing the chemistry Nobel prize.
A birth in crisis
Turning the first prototypes – flammable, explosion-prone safety hazards – into today’s omnipresent power source wasn’t always straightforward.

In the late 1960s, petrol production hit unprecedented heights. The US reached peak oil production in 1970, which started to put upward pressure on oil prices. Exxon, one of the largest oil and gas companies, noticed this trend and started recruiting scientists to work on alternative energy applications. Whittingham was one of those recruits.
He joined the company in 1972 from his post-doctoral fellowship at Stanford University in California, US, where he had been investigating intercalation – the reversible insertion of species such as ions or molecules into layered materials. At Exxon, Whittingham started searching for materials that could intercalate lithium ions and that he could pair with lithium metal in a battery.
Chemists had long been considering lithium as an element to use in batteries. Lithium is lightweight, which would give it huge advantages over lead–acid batteries. The prevalent batteries at the time, lead–acid, date back to the technology’s early days in the mid-1800s, when it was one of the first rechargeable batteries.
Lead–acid batteries are cheap, which is why they are still used in cars. But not only are these batteries heavy – not surprising given they contain lead electrodes – they are also dangerous. Excessive charging breaks down the dilute sulfuric acid that serves as the electrolyte, producing hydrogen and oxygen – an explosive mixture if the gas safety valve fails. And there are environmental concerns associated with mining and using lead, as the metal can form excessively toxic compounds.
The other rechargeable batteries widely available in the 1970s were nickel–cadmium, developed just before the start of the 20th century. However, they could only produce around 1.3V per cell. Lithium’s extremely low potential of –3V versus a standard hydrogen electrode held promises of much higher voltages.
But lithium is also reactive, almost too eager to give up its electrons, and taming it became a major problem. Aqueous electrolytes are a no-go since lithium reacts violently with water. And there is another reason: at more than 1.5V, water is split into hydrogen and oxygen – a safety problem that had already become apparent in lead–acid batteries. If the lithium battery were to produce almost twice that voltage, chemists had to find a different electrolyte.
Titanium disulfide has a layered structure that can reversibly accommodate lithium ions
By the end of the 1950s, PhD student William Harris at the University of California Berkeley in the US identified propylene carbonate as the most promising candidate.2 Though many modern lithium-ion batteries contain semisolid polymer electrolytes, carbonate esters remain common to this day.
But a suitable cathode material to pair with the powerful lithium anode was still missing. Researchers came up with a vast number of possible cathodes, though most of them only made for primary – non-rechargeable – batteries. Some, like lithium–iodine, are still in use because of their long lifespan of more than 10 years, which makes them useful for medical devices like pacemakers.
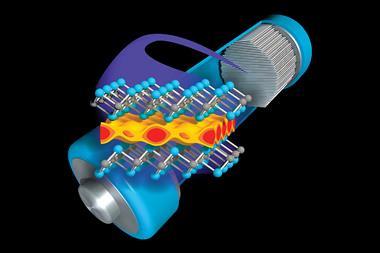
For a secondary (rechargeable) battery, the cathode would need to be able to take up lithium ions during use and release them again while being charged. Whittingham found such a material in titanium disulfide. The golden solid has a layered structure that can reversibly accommodate lithium ions. It is electrically conductive, doesn’t react with or intercalate any other species (like electrolyte molecules), and works under ambient conditions.
Firefighters on call
In 1973, at the height of the oil crisis that saw the price of a barrel increase by nearly 400%, Whittingham was ready to put titanium disulfide to work in a battery. ‘Exxon started a project in electric vehicles, they wanted batteries, and it was my job to see what would be feasible,’ Whittingham said in a news conference after the prize announcement. ‘So we came up with an idea for batteries. I had to go to New York and present it to a committee of the board. They said “Sounds good to us!” and within a week they were funding a major project.’
Whittingham’s design – a lithium-metal anode, a titanium disulfide cathode and lithium perchlorate in dioxolane as electrolyte – could be charged and discharged thousands of times without suffering performance losses.3 And most importantly, the battery put out an astonishing 2.5V. ‘The main advantage was an almost order of magnitude higher energy density,’ Whittingham explained. ‘Lithium-ion stored about 10 times as much energy as lead–acid and about five times as much as nickel–cadmium, either per unit weight or per unit volume.’
But despite initial excitement, the battery proved to have a major safety issue, as the team working on the battery’s commercial design soon found out. During charging, lithium isn’t deposited homogeneously on the anode. Instead, it grows into spiky structures. Over the course of hundreds of charging cycles, these dendrites can grow all the way across to the anode–cathode gap, dangerously short-circuiting the battery. The battery heats up dramatically, igniting the flammable electrolyte and eventually the lithium.
Lithium-ion stored about 10 times as much energy as lead–acid
Firefighters had to put out several blasts at the Exxon laboratory and threatened to start charging them for the special chemicals needed to extinguish burning lithium. Nevertheless, in 1976, Exxon began manufacturing the first rechargeable lithium battery. It contained aluminium in the anode, which slowed down dendrite formation and made the battery marginally safer.
Nevertheless, safety problems persisted. As oil prices went down after the mid-1970s crisis, Exxon withdrew the battery from the market and eventually shut down its battery research entirely.
Better than good enough

As the oil crisis and Exxon’s interest in batteries came to an end, Goodenough was only just getting started. In 1976, he was appointed head of the University of Oxford’s inorganic chemistry laboratory – a controversial choice, says Russell Egdell from the University of Oxford, who was doing his PhD research at the university at the time.
Although Goodenough had worked on energy storage materials at MIT’s Lincoln Laboratory in the US, he had never received formal chemistry training. ‘There was a sense of scandal in the inorganic chemistry community outside Oxford when an American physicist was appointed,’ Egdell says.
For the university’s undergraduate chemists, Goodenough’s lectures proved impenetrable. ‘He taught what was in retrospect a truly amazing course,’ Clare Grey remembers. Now a professor at the University of Cambridge, she is a former Goodenough student. Students started dropping out of the lectures, says Grey, so ‘a group of us decided that a better audience was needed. We found all the cuddly toys we could amass and filled rows two and three of the lecture theatre up.’ In hindsight, Grey says she is embarrassed. ‘But more than 30 years later we can both laugh about it’. ‘The teddy bears just managed to stay awake,’ Goodenough joked in an interview with the Nobel foundation.
The breakthrough in Goodenough’s battery research came in 1980. Using Whittingham’s titanium disulfide as a starting point, Goodenough made an educated guess: a compound with a smaller, more electronegative element than sulfur – namely oxygen – would make lithium intercalation energetically more favourable and give a higher cell voltage. Goodenough, says Egdell, was already known for his impressive ability to sketch out phase diagrams and predict regions of superconductivity – and he turned out to be right on this account, too.
Koichi Mizushima from Toshiba Corporation, Japan, who was working with Goodenough at the time and is a co-author on his seminal paper, started systematically screening metal oxides. ‘Although there were no layered oxides with a similar structure to layered sulfides such as titanium disulfide, I found there existed ternary layer compounds LiMO2 (M = V, Cr, Fe, Co and Ni),’ he explains. ‘I expected high voltages and high ion-mobility especially in [the cobalt and nickel compounds], because the d-orbitals of Ni3+ and Ni4+ and Co3+ and Co4+ ions were energetically deep and hybridisation with oxygen’s p-orbitals was large.’
After about a year, Mizushima could finally confirm lithium cobalt oxide to be the best of the bunch.4 The dark blue solid contains anionic sheets of oxygen ions and cobalt, sandwiching layers of lithium cations. The structure is stable, even as lithium ions move in and out during charging and discharging. Moreover, against a lithium-metal anode, Goodenough’s cathode achieved an astonishing 4V, almost doubling the potential of Whittingham’s battery.
Lithium cobalt oxide remains one of the most popular cathode materials to this day. ‘I never thought that [it] was a really important material,’ Mizushima says. ‘I thought better cathode-materials would be found near future at the time.’
While Goodenough’s battery still used a lithium-metal electrode, it was a decisive step towards the final design. But market-ready batteries were still almost a decade away.
‘[Goodenough] and I came up with lots of novel materials and ideas but American industry isn’t willing to invest 10 years’ worth of money into making a product,’ said Whittingham. ‘And I think Japan at that time was willing to do that.’
Coke is it
‘There was a lot of R&D on portable electronics in the 1980s, and so small and lightweight batteries, with high energy density and rechargeability, were needed,’ Yoshino said in an interview with Asahi Kasei in Japan, where he was working when he performed his prize-winning research – and still is. ‘The big buzzword at first was “portable”, soon joined by “cordless” and “wireless”. I just sort of sniffed out the direction that trends were moving in.’

Yoshino got to work on finding a new anode, which he wanted to pair up with Goodenough’s lithium cobalt oxide cathode. Because one thing was clear: the lithium-metal anode and its fiery reactivity had to go. Yoshino focused on a system with a different charge–discharge mechanism called the ‘rocking chair’ battery.
In the first lithium-metal batteries, the ions moving into the cathode during use came from metallic lithium being oxidised. In rocking chair cells, both electrodes can intercalate ions; one acting as lithium source, the other as lithium sink. The new battery would contain lithium ions but no metallic lithium and would therefore not grow the dangerous metal dendrites.
The rocking chair principle had been demonstrated as far back as 1938, in a battery that could shuttle bisulfate ions between two graphite electrodes. But scientists trying to simply replace the metallic anode in early lithium systems with graphite found that their batteries didn’t last long. The graphite layers could intercalate not only lithium ions but also electrolyte molecules, which led to the material breaking down over time.
We take for granted batteries that can be cycled thousands of times
Starting in 1981, Yoshino’s team investigated first conducting polymers and then various carbon materials, like carbon nanofibers and, in 1985, petroleum coke.5 The amorphous oil byproduct proved to be a winner. It was stable in the electrolyte (lithium perchlorate in propylene carbonate) and could quickly and reversibly intercalate lithium ions. Shock tests – which included dropping a piece of iron on the battery – showed the design to be significantly safer than lithium-metal batteries, which tended to explode under the same conditions.
For the next six years, Yoshino and teams at Sony and Asahi Kasei wrestled with engineering challenges. ‘The biggest issue in commercialising the lithium-ion battery was the design of the electrode,’ says Yoshino. ‘The carbonaceous material is a powder, lithium cobalt oxide is a powder. One of the most challenging issues was how to prepare and design the electrode with these powders and the current collectors, which are metal foils – copper and aluminium.’
‘The engineering component has really improved the performance,’ says Louis Piper, who works with Whittingham at the NorthEast Center for Chemical Energy Storage at Binghamton University. ‘We take for granted batteries that can be cycled thousands of times. It’s one of the things that’s underrated, but it’s what you build a society and an economy on.’
The mobile revolution
In 1991, Sony finally released the first commercial lithium-ion battery. With a charging voltage of around 4V and a specific energy of 80 watt hours per kilogram, the design was lighter and more energetic than anything else on the market and soon became competitive. It set the stage for the upcoming mobile revolution.
Over the last few decades, incremental improvements have almost tripled lithium-ion batteries’ energy density. ‘There were many kinds of improvements, but the basic design and the basic concepts of lithium ion batteries did not change,’ says Yoshino.
None of the laureates could foresee the enormous impact their work would have. ‘I really couldn’t anticipate it at all, it came as a surprise,’ Goodenough told Nature. ‘I’m very pleased that it’s something that is useful for society and brings people together.’
‘As far as I can tell, the amount of lithium-ion batteries is going to increase exponentially,’ said Whittingham. Indeed, Benchmark Mineral Intelligence forecasts that worldwide production will be at 174GWh in 2020, up from 28GWh in 2016. ‘99% of me could not imagine such a huge market for lithium-ion batteries,’ says Yoshino. ‘Only 1% could.’
But there are still problems to solve. ‘The lithium-ion battery is pretty good, but it has its problems, too,’ Goodenough said in a news conference once he had returned to Austin. ‘You can’t charge too fast, you can’t overcharge and you can’t do quite a few things.’
‘The issue we’re all addressing right now is getting the cobalt out,’ said Whittingham. ‘Most of the cobalt comes from the Democratic Republic of Congo and 30–50% of that uses child labour. There’s a huge drive on everybody’s part to remove as much of the cobalt as we can out of all these materials.’ While there are already some cobalt-free alternatives – like the lithium manganese oxides Goodenough, Michael Thackeray and others developed in the 1980s – they usually don’t have the same capacity.
‘One of the things I find quite amusing is the fact that the original battery that [Whittingham] made used lithium metal and one of the directions that batteries are now definitely going is replacing the carbonaceous anode with lithium metal again,’ says Piper. Theoretically, lithium–oxygen systems have three times higher energy density6 but putting them into practice proved difficult, says Shao-Horn. ‘What is really challenging is making the system reversible, largely as a result of instability of the electrolyte,’ she points out.
One way to address the hazards that come with a metallic lithium anode is to use solid electrolytes, like the barium-doped lithium glasses Braga’s and Goodenough are working on.7 ‘Besides being a very good ionic conductor as a good electrolyte should be, it also has a huge dielectric constant,’ explains Braga. ‘This means that it can polarise spontaneously, which allows the devices that are made with this electrolyte to store extra energy as electrostatic energy, as in a capacitor.’ The team is already in conversation with several companies over developing the energy storage device commercially.
Scientists are also looking into entirely lithium-free alternatives, such as sodium-ion and potassium-ion systems. Compared with lithium batteries, they would be cheaper and safer, though would also have lower energy density. But Yoshino says that it will take a long time to develop and commercialise an entirely new battery system.
I’m in the lab every day, I’m still working
John Goodenough
For now, lithium-ion batteries are here to stay. ‘Lithium-ion is such a beautiful technology, it has the advantage of being incredibly versatile,’ says Thackeray. ‘I’m more optimistic about getting incremental advances from lithium-ion before we find an alternative, superior system.’
Grey hopes that the prize will get everyone to appreciate the complex chemistry that powers their portable devices. ‘Many people treat batteries as black boxes that electrons come out of,’ she says. ‘Most chemical processes aren’t reversible, yet we have this highly engineered and optimised device with materials that can be cycled so many times outside their thermodynamically stable window.’
Whittingham believes that the recognition that comes with the Nobel prize will bring greater visibility to the field. ‘I think it passes on the message that we can have a cleaner and nicer world, and perhaps start to solve the climate problem,’ he said
The three laureates certainly aren’t running out of ideas yet. ‘I’m in the lab every day, I’m still working,’ Goodenough told Nature. ‘What would I do, just retire and wait to die? No, I don’t think so.’
References
1 T Placke et al, J. Solid State Electrochem., 2017, 21, 1939 (DOI: 10.1007/s10008-017-3610-7)
2 W S Harris, Electrochemical Studies in Cyclic Esters, PhD thesis, University of California, Berkeley, 1958 http://bit.ly/2IZlSJj
3 M S Whittingham, Science, 1976, 192, 1126 (DOI: 10.1126/science.192.4244.1126)
4 K Mizushima et al, Mater. Res. Bull., 1980, 15, 783 (DOI: 10.1016/0025-5408(80)90012-4)
5 A Yoshino, K Sanechika and T Nakajima, Japanese patent no. 1989293, 1985
6 D Oh et al, Nat Commun., 2013, 4, 2756 (DOI: 10.1038/ncomms3756)
7 M H Braga et al, Energy Environ. Sci., 2017, 10, 331 (DOI: 10.1039/c6ee02888h)












1 Reader's comment