Putting the crunch into those moreish goodies

Karl Fischer: German chemist (1901–1958). Inventor of a simple system for determining trace water content in anything and everything from petroleum to crostoli (right)
There are some words that are untranslatable. English speakers love the German word schadenfreude. But Italian has words like sprezzatura (artful effortlessness) while Turkish has üsenmek (not having the energy to do something). In English, my favourite is the adjective moreish, an 18th century neologism used to describe a food that one cannot resist eating. And eating. And eating.
At the heart of moreishness is the texture, a quality often overlooked in Western cooking but which, as the food writer Fuchsia Dunlop points out, is central to Chinese cuisine. For the Chinese, crunchy, squishy and glutinous are as fundamental to food as bitter, sweet and sour are to us in the West. And much of the pleasure of crunch proves to be related to the sound, transmitted via our jawbone to the ear; the most moreish of foods are often the loudest. Each of us has our favourite: peanuts, chevda, keropoks, crisps, crostoli, meringues… the list is almost endless.
For the food manufacturer, the key to maintaining crunch is rigorous control of moisture content. So its determination is one of the fundamental analytical procedures in the quality control process. At its heart lies a simple chemical reaction originally discovered by Robert Bunsen, but that was exploited almost a century later by a German chemist working in the petrochemical industry.
Joseph Karl Anton Fischer was born in Pasing, a suburb of Munich, in 1901. A keen experimenter, as a child he had a laboratory in his bedroom where, among other things, he once prepared nitroglycerine. Although it burned satisfyingly, the mixture failed to detonate because his product was heavily contaminated with glycerol.
He was also very keen on microscopy and built his own microtome to prepare thin sections using a razor blade – he was so good that he was able to make a little money for his family during the difficult years of hyperinflation in the 1920s.
He studied at the University of Leipzig and his PhD, awarded in 1925, was on the effect of magnesium oxide on the vulcanisation of rubber. He then went to work in industry, finding a job with the company founded by Lazar Edeleanu, a Romanian organic chemist who first prepared amphetamine as part of his PhD. Edeleanu had invented a process in which liquid sulfur dioxide was used as a solvent to separate aromatic hydrocarbons from the aliphatics. By the time Fischer joined the company, sulfur dioxide refining was spreading across Europe.
It is not clear why Fischer started doing analytical chemistry, but water is a massive problem in the oil industry and its presence can lead to the formation of intractable emulsions. Ernest Dean and David Stark had developed their clever distillation method for breaking the emulsions, but the method only worked for large quantities of water. If the percentage were lower, something more subtle was required. Working for Edeleanu, sulfur dioxide would quite literally have been in the air. What Bunsen had spotted in 1853 was that SO2 could be oxidised to sulfuric acid by iodine in the presence of water.
SO2 + I2 + 2H2O ? H2SO4 + 2HI
Adding a suitable base like pyridine or aniline, Fischer found that the equilibrium was driven strongly to the right. In his 1937 paper, Fischer proposed a breathtakingly simple titration using a standard methanolic solution of iodine, sulfur dioxide, and pyridine, where the end point was signalled by the persistence of the brown iodine. Neither Fischer nor his company took out a patent and the method entered the public domain. Although the stoichiometry would be revised two years later, it was widely adopted.
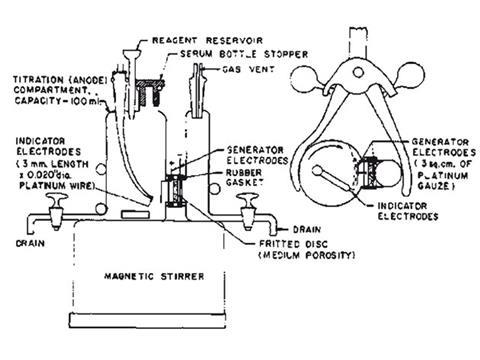
But it required a manual titration. In 1959, two electrochemists at the Oak Ridge National Lab, US, reasoned that if iodide could be converted to iodine with 100% current efficiency at a platinum electrode in methanol, just as it was in water, then the total electric charge transferred would be proportional to the amount of water. A two compartment cell was constructed with a platinum gauze electrode to generate the iodine from a depleted Fischer reagent, and the end point detected by the ‘dead stop’ method: two identical platinum electrodes were placed a few millimetres and a few millivolts apart. When the water was used up, a current surge between the two signalled the end point.
Robust, extremely sensitive down to ppm levels, automatic and very fast, the coulometric Karl Fischer method for water was soon being marketed as a complete kit, using imidazole in place of the smellier pyridine. Fischer, however, never saw this. After leaving Edeleanu he worked as an itinerant chemist as well as an academic. He died of a heart attack in 1958.
Today the KF titration is applied in every industry where water matters. Everywhere, in other words. When you next crack open a packet of grissini, macadamias or digestive biscuits, don’t just think KF. Think of the word ‘crunch’ – it isn’t as easy to translate as it sounds.
Andrea Sella (@SellaTheChemist) teaches chemistry at University College London, UK











No comments yet