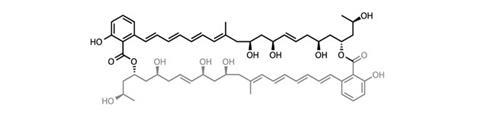
From a synthetic point of view, oligo- and polymeric molecules at first glance look rather simple – after all, the repeating unit need only be synthesised once. But controlling the amount of repetition is far from trivial, especially in the case of cyclic dimers such as marinomycin A. However, a team led by Andrew Evans at University of Liverpool, UK, has a neat solution to this problem – starting with their decision of where to split the dimer.2 The team had several options. For example, ring closing metathesis provides a great opportunity for splitting across olefins. But they chose the flexibility of carbonyl chemistry and the ester groups around which to split the target. As most undergraduates learn early in their chemical tuition, carbonyl groups are extremely tunable in their reactivity, a feature that the team planned to employ in some selective dimerisation chemistry.
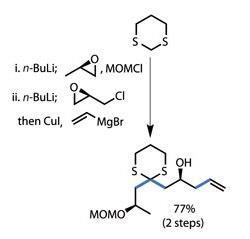
Freeing the carbonyl group with iodine delivered a rather complex fragment in only a few steps. The team then went one stereocentre further, reducing the freed carbonyl diastereoselectively with samarium diiodide to complete the first fragment.
The team coupled this with a second fragment, made in five steps from a d-ribose derivative, and a further unsaturated moiety finishing in a salicylic acid group to finish the monomer (figure 2).
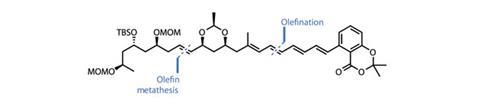
On the other hand, making an acetal between the carboxylic acid and phenolic hydroxyl eliminates that hydrogen bond and makes a much more reactive ester. By combining the two, the team completed their first ester stitch to form a dimeric intermediate, with a pleasing 82% yield. They then reactivated the other salicylic acid group by protecting the phenol to disrupt the hydrogen bond, allowing a speedy and effective intramolecular cyclisation reaction to complete the macrocycle in a reasonable 34% yield (figure 3).
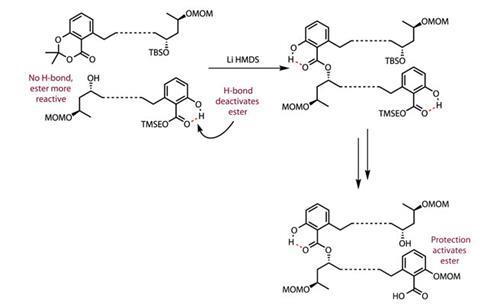
What is even more interesting is what this may imply about the biosynthesis of such molecules, as once again it appears nature may have got to this strategy first.
Paul Docherty is a science writer and blogger based in Reading, UK
References
- H C Kwon et al, J. Am. Chem. Soc., 2006, 128, 1622 (DOI: 10.1021/ja0558948)
- P A Evans et al, Nat. Chem., 2012, DOI: 10.1038/nchem.1330
- A B Smith and M Xian, J. Am. Chem. Soc., 2006, 128, 66 (DOI: 10.1021/ja057059w)












No comments yet