Chris Nawrat
Chris Nawrat obtained his PhD in total synthesis in the UK, completing several brightly coloured natural products. After a postdoctoral stint at an Ivy League school, he remained in the US, where he now works as a process chemist at a major pharmaceutical company.
 Opinion
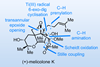
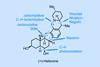
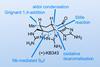
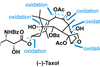
Opinion(+)-Melicolone K
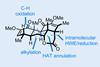
Sequential C–H activations open up the opportunity for an unusual transformation
 Opinion
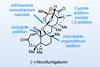
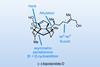
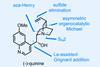
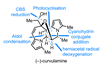
Opinion(−)-Novofumigatonin
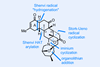
Oxidations abound in this satisfying synthesis, with a delicate nitrile hydrolysis to finish
 Opinion
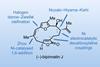
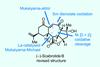
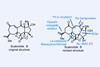
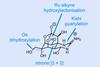
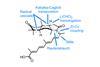
Opinion(–)-Scabrolide B (again!)
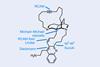
Proverbially, comparison may not bring joy – but it can be educational
 Opinion
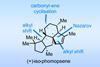
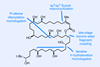
Opinion(+)-iso-Phomopsene (and friends)
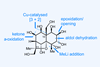
A non-obvious combination unleashes the power of electrocyclisation
 Opinion
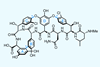
Opinion(±)-Rhabdastrellic acid A
A triterpene that looks innocuous until it reveals the highly strained conformation of its rings