How does an organism without a brain or a nervous system sense when it’s under attack? Hayley Bennett presents the plant world’s strange yet sophisticated system for responding to wounding
Being rooted to the spot, plants don’t have the luxury of running away when they’re attacked. They do have a system to deal with being eaten, but it doesn’t revolve around nerves or a brain. Say a caterpillar is munching on a leaf; the plant immediately senses the damage and mounts a response, diverting its resources from growth to defence and, for example, churning out combat chemicals destined for the caterpillar’s guts. This defence system is triggered by chemical and electrical signals, transmitted surprisingly rapidly, and their exact mechanisms have puzzled plant scientists for decades.
Faced with a lack of facts about how precisely these fast-acting plant defences operate, some outside the field have tried to claim that they must use similar mechanisms to the impulses of our own nervous systems. Plant neurobiologists would have us believe that the mechanisms driving wound signalling in plants somehow mirror the processes that animals use to transmit nerve signals. But according to David Robinson, a plant scientist at the University of Heidelberg in Germany who has refuted the theory in several papers, ‘The basic desire [of plant neurobiologists] is to have people believe that plants and animals are fundamentally alike, so that plants can think, make decisions, have memories, feel pain… Frankly, the evidence for these claims does not exist.’

So if plants don’t have nerves, or a brain, then how do they register being eaten? That’s what’s so interesting, according to Ted Farmer, a plant signalling expert at the University of Lausanne in Switzerland. Despite a background in animal biology, Farmer says he’d be ‘very sad’ to find the same mechanisms in both plants and animals. ‘[Plants] are strange organisms that are completely unlike us,’ he says. What he and others are now uncovering is a separate, but still sophisticated, system for sensing the world that enables plants to respond rapidly to wounding and other stresses, even in the absence of nerves. It may have taken them over a century, but the science of plant signalling is in full bloom.
Rapid response
One plant whose surprisingly speedy responses have long fascinated scientists – and amateur gardeners – is the ‘shameplant’ or ‘touch-me-not’, Mimosa pudica. The shameplant’s feathery leaves react immediately to touch by folding up, apparently as a defence against hungry herbivores. A century ago, Italian plant scientist Ubaldo Ricca tried to work out what was driving these movements, though he had access to only the most rudimentary of apparatus: some red dye, a stopwatch and a pair of scissors. Ricca would cut a stem of Mimosa, dip it into the dye and then, as its leaves began to curl, cut the stem further up to see how far the dye had travelled. By the time a leaf had curled up, the dye had almost always reached it. This worked even with dead plants, suggesting that Mimosa relied on simple diffusion of some unknown chemical substances – dubbed Ricca’s factor or factors – to transmit a warning signal through its water-carrying tubes, or xylem. This thinking was later expanded to wounding in plants more generally.
In the 100 years since, the plant science community has improved on Ricca’s experimental set-up somewhat, coming at the problem armed with modern imaging techniques, genetics and scissors (even today), but without reaching a very satisfactory solution. ‘There have been people who have shown data that has very much fallen in line with [Ricca’s work] – the concept of a mobile molecule or set of molecules that might initiate responses – and there are others who’ve said it’s just completely implausible,’ says Christine Faulkner, a plant researcher at the John Innes Centre in Norwich, UK, who has been studying wound responses in the model plant Arabidopsis (rock cress).
Besides straightforward diffusion of molecules released in wounds, modern theories cover active mechanisms requiring the machinery of living cells and hydraulic waves initiated by water pressure changes when plants are damaged. Models for plant wounding have become more and more complex as scientists have tried to combine multiple possible mechanisms, leading to journal papers containing diagrams with ‘arrows everywhere, to everything’, Faulkner says.
Meanwhile, researchers have also been sticking electrodes on plants to measure electrical signals that shoot through stems and leaves following wounding. It’s not certain what causes these either, but as Farmer explains, chemical and electrical signals probably originate at least partly from the same source. ‘Plants are chemical factories,’ he says, and when their cells are wounded, the compound mix that spills out inevitably contains some ions whose movements alter the balance of electrical potential across cell membranes. If this change in membrane potential was somehow transmitted from one cell to the next perhaps it could produce an electrical signal.
A hidden world
One ion that both chemical and electrical approaches converge on is the calcium dication (Ca2+ ), which is known to be shunted across membranes into plant cells after wounding – in distant as well as wounded leaves. Since the 1990s, plants genetically engineered to make their own calcium indicators have uncovered what has been referred to as a ’hidden world’ of calcium ions in plants. Revealing more than Ricca’s dye ever could, plants engineered to make a bioluminescent jellyfish protein (aqueorin), for example, glowed blue when this protein bound calcium. In 2015, German researchers used this technique to watch calcium fluxes across whole plants as they were being munched by moth larvae. They clearly saw calcium levels spiking in leaves other than the ones being chewed, suggesting calcium was involved in transmitting the wound signal from leaf to leaf.
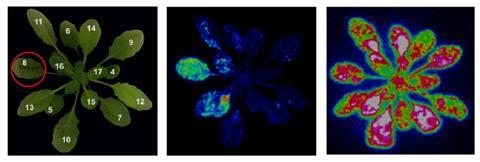
Calcium is a secondary messenger, so it’s not the first link in the chain (and probably not the second either), but work on calcium waves in plant cells has provided a bunch of beautiful-looking images that have convinced scientists that it is key to wound responses. The focus on calcium is partly because it is easy to visualise, according to Alex Costa, a plant physiologist at the University of Milan in Italy. Cells keep calcium concentrations much lower on the inside than the outside – to stop salts forming – so any sudden inflow results in a strong signal. In this respect, imaging calcium in a cell is like working in a darkroom, until just a few ion channels open up. ‘In a darkroom,’ says Costa, ‘even if you switch on a small and very tiny light, it can be easily visible.’ Because the concentration gradient between the inside and outside is so steep, the cell becomes very quickly flooded with calcium – and light.
So calcium became one piece of the puzzle, but meanwhile Farmer’s group was trying to work out how others fitted. The team had gone looking for genes required for plant wound signalling and in 2013 they became the first to identify one of the genetic components of the pathway. Their discovery ‘opened up this field of research’, Costa says. Their results showed that plants lacking working glutamate receptor-like (GLR) genes produced weaker electrical responses when injured with forceps. These plants were also less able to activate the plant defence hormone jasmonate, which normally starts being made within minutes of a caterpillar attack. Perhaps most intriguingly, though, GLR genes encoded proteins acting as cell gateways for the central protagonist of the wound signalling story: calcium.
Even today, though, calcium’s role remains mysterious; it’s not clear what information it’s carrying, although it’s thought it may link to jasmonate activation. ‘The calcium, in my mind, is probably the most complicated part of the story,’ says Farmer. ‘What’s it actually doing? I think we know less than we think we know about the calcium.’ Costa, meanwhile, points to the 250 different calcium-binding proteins in plant cells, which suggest that when calcium gets in, it almost can’t help but activate a response, even if we don’t understand what, exactly, that is yet.
Still, upstream of calcium, there was now more to go on following Farmer’s discovery. The realisation that GLRs were involved suggested a role for the amino acid glutamate – in animal cell membranes, there are protein gates equivalent to GLRs for which glutamate acts as a kind of key, allowing calcium to rush in to cells. Could glutamate and calcium be linked in plant wound signalling too?
Connecting the dots
In 2018, another impressive feat of imaging from a team led by Masatsugu Toyota at Saitama University in Japan answered the question. Their approach demonstrated again the power of fluorescent reporter molecules for visualising plant responses at the larger scale. ‘You can just use microscopes to magnify the small world, but we weren’t curious about [single] cells,’ says Toyota. ‘We wanted to visualise the calcium response in entire plants.’ Employing the next generation of genetically encoded jellyfish reporter proteins, they observed calcium and, for the first time, glutamate signals wash across rockplants in response to their leaves being cut or crushed. Glutamate, they suggested, was released when cells were damaged and was involved in wound signalling too.
Toyota’s work connected some dots, linking calcium waves to glutamate and to the GLR ion gates – plants without these cellular gates didn’t light up when wounded. ‘That was a big Science paper and everyone was like “Wow!”’ says Faulkner. But as glutamate was only seen to spread locally it wasn’t viewed as the primary signal. At this point, most plant scientists looking for some sort of mobile messenger had also ruled out calcium (for one thing, it reacts so readily that it wouldn’t get very far). And there was still disagreement about whether the wound signal could really be carried by molecules simply diffusing, as Ricca proposed. Many scientists thought it required a pressure wave or active mechanism for cell-to-cell transport, potentially involving the calcium.
We could see that the wave was slowing down over time
So when the John Innes Centre team set about using the same fluorescence reporters as Toyota’s group to tackle the problem themselves, it was with Ricca’s theory in their ‘back pocket’, recalls Faulkner’s PhD student Annalisa Bellandi, now based at École Normale Supérieure de Lyon in France. Bellandi had a key insight about the calcium wave that allowed them unpick the transport mechanisms. Visualising it as a ‘doughnut wave’ that spreads from an injury site, she devised new data analysis techniques to measure not just the fluorescence inside the doughnut – as others had done – but to track the speed of the calcium wave front as the doughnut grew in size. ‘Then we could see that the wave was slowing down over time and this is something that the other method could not see,’ she says. Based on this, in their 2022 paper, they suggest that whatever is triggering the calcium wave, it isn’t being actively propagated through leaves and veins – in that case, the speed of the wave front would have remained constant. Instead, it was diffusing.
‘What was really satisfying for us,’ Faulkner says, ‘is that, really, those guys that were doing all of these experiments way back – probably working with what was a very rudimentary stopwatch and some food colouring – kind of had the idea right.’
What’s more, the team’s detailed analysis of calcium and glutamate waves, combined with mathematical modelling, gave new insights into their transport that placed glutamate back under the spotlight. Their model casts glutamate as a leading Ricca’s factor, which is released in damaged leaves and diffuses through neighbouring plant cells along the passages that link together their cell walls. The calcium wave, meanwhile, is relegated to a ‘read-out’ of glutamate busting open the GLR cell doors as it passes through. When a big vein is severed, they suggest, glutamate also gets into the xylem vessels, which transport it to distant leaves via water flow. If correct, this model offers a simpler model for wound signalling than some might have expected.
The bigger picture
But this new version of events poses yet more questions. Toyota, for example, isn’t yet convinced that a glutamate signal can explain very long-distance signalling. ‘How glutamate can transfer at a very fast speed from the wound site to a distant site, we have no idea.’ In recent work, he applied modern techniques to Ricca’s original test subject – the shameplant – observing fluorescent calcium signals and electrical signals travelling in tandem to trigger leaf-closing within seconds of touching the leaves. He says that in this case it’s still unknown what drives the calcium signal so fast.
As Costa points out, Faulkner’s results centre on glutamate signals travelling from one leaf to the next in seedlings. But his own team’s latest experiments also show a rapid increase in glutamate that occurs at distant sites, in larger plants. Interestingly, they saw this glutamate signal even in the absence of any cutting or crushing; when they used sorbitol to draw water from the roots of hydroponically grown rockcress, creating osmotic stress, they still saw a response in leaves and what would become the main flower head. So what Costa is suggesting is that the primary signal is actually a change in water pressure inside cells, while glutamate opening the calcium gates is what happens further downstream, in distant parts of the plant.
In our model, it’s not just diffusion
It seems likely that plants are, in fact, operating more than one system, with the leaf-to-leaf and root-to-leaf systems potentially differing. While Farmer likes the John Innes team’s work ‘very much’ and agrees that it can ‘explain an awful lot about triggering the calcium signal’ near to a wound, he also believes that water fluxes and pressure changes may have an important part to play in longer distance signalling. So, while glutamate and other Ricca’s factors may spread locally near to wounds, his most recent model emphasises how these chemicals draw water out of cells by changing their membrane potential. In this model, the water fluxes help to ‘siphon’ Ricca’s factors or what Farmer calls ‘elicitors’ into the water-carrying xylem vessels, so they can be transported further away. Meanwhile, waves of membrane depolarisation are linked to fast electrical signalling. ‘So in our model, it’s not just diffusion,’ he says. ‘It’s a bit more complicated.’
As Faulkner says, the mechanism they’ve uncovered is probably only one piece of a much bigger puzzle – there are probably many more factors involved in plant signalling, and response, just waiting to be discovered. But unlike for calcium and glutamate, she says, ‘We have no sensors. So we have to start at the beginning again.’ Plants, it turns out, don’t need to have brains to be complicated.
Hayley Bennett is a science writer based in Bristol, UK













No comments yet