Fifty years after its birth, John Mann reports on the conception and evolution of the contraceptive pill
In Mexico City in 1943, the small company Laboratorios Hormona occupied a niche in the market for pharmaceuticals. Its founders, Hungarian Emeric Somlo and German Federico Lehmann, were making a modest living selling natural hormones extracted from animal organs.

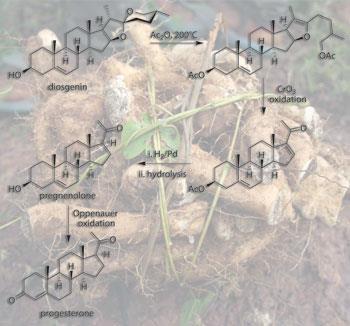
Imagine their surprise when one day a rather eccentric American professor of chemistry called Russell Marker arrived in their office bearing 2kg of pure progesterone worth around $160 000 (£107 000) at that time (many tens of millions of dollars at today’s prices). The high cost of this sex hormone, and others such as testosterone and oestradiol, was due to the multistep syntheses required to make them. Marker had achieved what previously seemed impossible - a cheap five-stage synthesis of progesterone from the natural product diosgenin.
Marker makes his mark

Marker’s pivotal discovery arose from studying a class of plant natural products called sapogenins. He elucidated the structure of sarsasapogenin, from sarsaparilla root, and discovered that it had the same basic ring system as cholesterol but with an oxidised side-chain. He was intrigued by the possibility of using these oxidised sites to affect a cleavage of the side-chain and make the degraded structures of the sex hormones. This is, of course, exactly what happens in the enzyme-mediated oxidative metabolism of cholesterol to produce progesterone (see Chemistry World, November 2009, p54). In 1939 he devised chemistry that worked on sarsasapogenin but realising that this sapogenin was relatively expensive, he sought to carry out similar chemistry on the more accessible diosgenin from the genus Dioscorea.
Being something of a botanist as well as a chemist, Marker headed across the border into Mexico in search of Dioscorea mexicana – the Mexican yam that the locals called cabeza de negro. After considerable difficulties he found a good source near the city of Orizaba in Veracruz State, and transported large quantities of the yam back to his laboratories at Pennsylvania State College.
During 1942 he perfected his oxidative degradation to make progesterone from diosgenin in just five chemical steps. Unable to interest US pharmaceutical companies in his process, he headed back across the border and ended up at Laboratorios Hormona. Unlike their US counterparts, Somlo and Lehmann immediately recognised the novelty and merit of Marker’s work. The three men set up a new company, Syntex, in January 1944 to exploit the process. Initially Marker synthesised several more kilograms of progesterone, but then left Syntex after a number of disagreements. This left the company short of the necessary chemical expertise to continue his work. The gap was immediately filled by another Hungarian, George Rosenkranz. And four years later the chemist who has become known as the ’father of the pill’ – Carl Djerassi – also joined the firm.

Syntex rapidly became the leading supplier of sex hormones including progesterone, testosterone and various oestrogens, all made from diosgenin. Then, in June 1951, the company reported what was surely the highlight of their academic research - the 20-stage chemical synthesis of the most clinically important steroid of the era – cortisone. Previously this key anti-inflammatory steroid could only be made using a 32-stage synthesis pioneered by the company Merck & Co. The Mexican group completed their historic synthesis a few weeks ahead of the major research groups of both Robert Woodward and Louis Fieser at Harvard University, US – a huge public relations triumph for Syntex.
Later they went on to improve their route by preparing cortisone from a very cheap sapogenin called hecogenin that comes from the sisal plant. This process was later licensed to the pharmaceutical firm Glaxo and formed the basis of that company’s world-leading steroid chemistry, leading to blockbuster drugs such as Betnovate (betamethasone) and Becotide (beclomethasone). With their expertise in steroid chemistry established, the Syntex group were now well prepared for the discoveries that were to cement their place in history.
The pill’s humble beginnings
The discovery of the contraceptive pill was, like so many seminal discoveries in medicinal chemistry, a mixture of serendipity and inspired chemistry. While Syntex was marketing progesterone as a drug to prevent miscarriage and to alleviate menstrual problems, its water insolubility and short half-life (about 5 minutes) in serum meant that oral delivery was impossible.
Two unconnected discoveries triggered the research undertaken by Djerassi and Rosenkranz. Firstly a group at pharma firm Schering in Berlin, Germany, had synthesised ethisterone (a testosterone analogue with an ethynyl group at C-17) in 1938, which had progesterone-like activity and was orally available. Even more importantly, in 1944 Maximilian Ehrenstein at the University of Pennsylvania in the US had synthesised a small amount of 19-norisoprogesterone from the natural product strophanthidin (an arrow poison from an African plant). Surprisingly this also exhibited progesterone-like activity (despite the lack of a 19-methyl group and inverted stereochemistry at C-14 and 17).

In 1951, Djerassi and Rosenkranz prepared a sample of 19-norprogesterone (with correct stereochemistry) from progesterone and confirmed that it was more active than progesterone. They then tackled the synthesis of a steroid that combined the loss of the 19-methyl group (19-nor-series) with an ethynyl group at C-17 by preparing 19-norethisterone (later called norethindrone), and demonstrated that it had greater progestational activity than any other known steroid and was orally active.
They filed for a patent in November 1951 and then described their research at an American Chemical Society conference in April 1952, a whole year before the US pharmaceutical company G D Searle announced norethynodrel. These two progestogens were destined to be the main constituents of the first contraceptive formulations.
Oestrogen’s role
Progesterone had long been associated with an anti-ovulatory effect as well as its ability to prevent contraction of the uterus thus helping to prevent expulsion of an embryo. The first demonstration that norethindrone and norethynodrel were effective as anti-ovulatory agents was revealed by Gregory Pincus of the Worcester Foundation for Experimental Biology in Massachusetts, US, and John Rock the Harvard-based clinician in 1956.
Almost by chance it was discovered that small amounts of the synthetic oestrogen mestranol were also present in samples of these new synthetic progestins and seemed to enhance the anti-ovulatory effect. As a result, the first contraceptive pill formulations contained a mixture of the 19-norprogestogens and small amounts of mestranol.
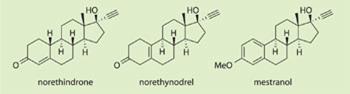
Extensive clinical trials took place in Puerto Rico (and to a lesser extent in Mexico) between 1956 and 1958, and the hugely positive results (only those women who withdrew from the trials became pregnant) led to US Food and Drug Administration (FDA) approval of Enovid (norethynodrel and mestranol) for contraceptive use.
In 1960, it was launched by G D Searle, and Ortho-Novum (norethindrone and mestranol) was released jointly by the firms Ortho and Syntex in 1962. Then in 1964, Syntex launched its low dose oral contraceptive pill Norinyl with each pill containing 2mg of norethindrone and 0.1mg of mestranol; and this kind of formulation has remained the mainstay of the pill ever since.
Male contraceptives
In China in the 1950s, it was found that using crude cottonseed oil for cooking seemed to be associated with an increase in male infertility. This led to evaluation of the oil’s major constituent gossypol in animal studies. These were successful and clinical trials on male volunteers began in 1972. Participants were initially fed daily doses of 20mg of gossypol until their sperm counts dropped below what was deemed necessary for procreation (usually less than 1 million sperm per ml of seminal fluid). The 4000 volunteers then received maintenance doses of 75–100mg twice monthly for periods of up to 4 years with a claimed 99.9 per cent success rate in terms of pregnancy prevention. It was also claimed that fertility completely recovered when the men stopped taking gossypol. However, this method of fertility control never became established.
As long ago as 1939 it was shown that testosterone propionate could cause a low sperm count (azoospermia). And in the most recent and extensive study, the Chinese have turned to the administration of testosterone undecanoate (500mg monthly). A multicentre Phase III study, involving 1045 men over a 30 month period, revealed a near complete and reversible result for azoospermia with a contraceptive failure rate of 1.1 per 100 men during this period. However, the trial’s leading scientist Yiqun Gu told Chemistry World that no further studies are under way.
Several other studies have used combinations of testosterone derivatives in conjunction with synthetic progestins, and while most of these combinations produced acceptable levels of azoospermia, there were usually unacceptable side-effects most importantly loss of libido.
Here we go round the mulberry bush
A further intriguing compound is under examination at the department of pharmacology at the University of Oxford, UK. The N-butyl derivative of the natural product deoxynojirimycin, found in mulberry leaves, has been of interest for at least 20 years, and initially showed promise as an antiviral agent. Now Frances Platt’s group has shown that the compound impairs sperm formation in mice. N-butyl deoxynojirimycin appears to render sperm unable to penetrate the female egg. Since fertility returns once the drug is no longer taken – and appears to have no harmful effects on body weight, the testes, liver and kidneys – its use in humans might be worth exploring.
The natural approach
A more natural form of male fertility control may arise from studies of gonadotropin-inhibitory hormone. This small peptide, produced by the hypothalamus, prevents the synthesis of FSH (follicle-stimulating hormone) and LH (luteinising hormone) in the pituitary gland. And once the balance between this inhibitory hormone and gonadotropin- releasing hormone are understood at the neural level, it may be possible to design small peptides that disrupt the delicate balance and therefore affect the production of oestrogens and testosterone. This research is at an early stage, and as one of the leaders in this area - George Bentley of University of California Berkeley, US – claims that ’in male rats the hormone reduces copulation felicitation, that is the rats lose interest, and this is not a desirable strategy for human use’. So there is clearly much to do before this type of therapy would be acceptable.
A booming market
By 1966 more than 5 million US women were using the new oral contraceptives. In the UK, the first prescriptions were written in 1961 with 480 000 users by 1964, though the pill was initially only available to married women and wider use only occurred when the Brook Family Planning Clinics opened in 1964.
Since the mid-1960s many new variants of the original steroids have been introduced, but the most successful strategies have changed little over the past 50 years. These include: a combined oral formulation of progestogen and oestrogen; an oral formulation containing only progestogen; and slow release formulations using an IUD (intrauterine device or a coil), transdermal patch, implantation or a vaginal ring.
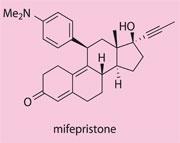
The only major innovation has been the introduction of the emergency contraceptive (actually an abortifacient) RU-486 or mifepristone in 1982, which acts by blocking progesterone receptors in the uterus.
There have been attempts to develop a non-steroidal drug for fertility control, but the only widely used drug is ormeloxifene which acts upon oestrogen receptors in cells of the uterus (and breast) to modulate gene expression in these cells. In this respect, the drug has a similar mode of action to the breast cancer drug tamoxifen. Ormeloxifene has been in use in India for around 20 years and has recently been introduced into South America, but there are no licences as yet for use in Europe or North America.
Finally, the potential use of so-called microbicides has been highlighted at a recent conference (Microbicides 2010, Pittsburg, May) on these interesting antimicrobial peptides. These compounds might be tailored to be both spermicidal and to offer protection against HIV and other pathogens. However, since they would probably have to be applied prior to sexual activity, they would not be as convenient as the contraceptive pill.
Reaping the benefits
Over the years there have been a number of scares concerning the potential for the pill to cause thrombosis, but these problems have been largely overcome through the use of low-dose progestogen-only formulations. In addition, there is a well substantiated long-term protective effect against both ovarian and endometrial cancers. In a March 2010 article in the British Medical Journal, that reviewed a study of more than 46 000 women over a period of 39 years, Philip Hannaford of the University of Aberdeen, UK, wrote: ’oral contraception was not associated with an increased long-term risk of death indeed, a net benefit was apparent.’
One of the main issues with the introduction of the contraceptive pill was the fact that men no longer needed to worry about insemination, meaning the responsibility lay solely with the women. This was partially overcome with the appearance of HIV and Aids in the 1980s and the urgent need to avoid unprotected sex. In addition, the development of the vasectomy and its reversal provided men with a means for controlling their own fertility, and in consequence there has been a limited ongoing search for chemical methods to control male fertility (see box above).
So after 50 years of use how can we rate the success of the contraceptive pill? It is currently used by an estimated 100 million women of childbearing age worldwide, though the percentage use ranges from less than 5 per cent in parts of Africa and Asia to as much as 60 per cent in the developed world. While its use has undoubtedly helped women to take control of their fertility, in those societies where for reasons of religion, social unrest or poverty the use is restricted, there are an estimated half million unplanned conceptions every day, many of which are terminated by chemical or surgical intervention.
As Djerassi wrote in his 2001 book One man’s pill : ’there is one categorical answer that can be given to the question of the pill’s benefits versus its risks, I am referring to the number of unwanted children and abortions – the sheer mass of human suffering – prevented over the years.’
John Mann is emeritus professor of chemistry at Queen’s University Belfast, UK. He worked as a postdoctoral scientist at Syntex, Palo Alto, US, during 1971












No comments yet