Experiments probing how force accelerates chemical reactions have provided new insights into mechanochemical kinetics. The findings by US-based scientists could inform the development of chemical manufacturing methods that are more sustainable and less wasteful than current approaches.
In mechanically activated organic chemistry, force drives the making and breaking of covalent bonds. This helps to minimise the waste and energy cost associated with organic synthesis because the reactions are run neat or with minimal solvent, and energy is provided through mechanical, rather than thermal, means.
However, as materials scientist and organic chemist Adam Braunschweig from the City University of New York explains, mechanochemistry has not yet been widely adopted, in part because of the substantial gaps that remain in our understanding of reaction kinetics during these reactions.
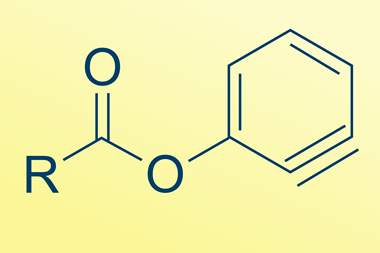
Aiming to provide answers, they experimentally and computationally studied the reaction kinetics of mechanically activated Diels–Alder cycloaddition reactions between four dienophiles and surface-confined diene monolayers to measure how force affects reaction rates.
The reactions were carried out on monolayers so that the complexities associated with grinding powders during milling and reactant availability were not factors in the kinetic analysis. This meant that the effects of force on the free energy of activation and reaction trajectories would be isolated.
The researchers then used elastomeric arrays containing 900 pyramidal tips to bring fluorescently labelled dienophiles into contact with monolayers of the tethered diene, anthracene, that was immobilised covalently onto the surface of a silica wafer.
‘We attached [the tips] to a piezo actuator, which just allows you to press very controllably into the surface,’ explains Braunschweig. ‘We pushed down, for one second, two seconds, three seconds, so we got the full-time course. Then, what’s great about these tips is you can tilt them with respect to a surface; so one bit touches with a different force than another bit.’
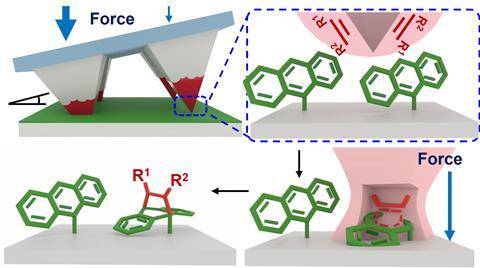
‘In a single experiment, you get the entire time course and all the forces plus each of these patterns is repeated 30 times, so you get really good, high fidelity, low-error data.’
They then used fluorescence microscopy to track the Diels–Alder adduct formation as a function of applied force and reaction time under force.
‘We discovered that the moderate mechanical energy that is applied along the right trajectory, can substantially accelerate the rate of organic reactions,’ adds Yerzhan Zholdassov, a doctoral student in Braunschweig’s lab and lead researcher on the study.
‘Moreover, different molecules have different responses to the applied mechanical energy,’ he says. ‘These findings have major implications in chemical industry, which allows us to produce chemical products without the harmful solvents and high energy input.’
It’s not just an extension of what you see in solution – something different is happening when you smash molecules at interfaces
Braunschweig highlights two ‘really surprising’ findings in the data they collected. ‘The first is that to get the reaction rate to increase tenfold, we only had to push with pressures of about three atmospheres. The other, really crazy, thing that we learned was that with Diels–Alder the activation volumes under force where you’re pushing in one direction [is around] 20,000 – a 1000-fold increase in the sensitivity to pressure when you’re using mechanochemical conditions [compared to hydrostatic pressure]. So it’s not just an extension of what you see in solution, it means something different is happening when you smash molecules at interfaces.’
Tomislav Friščić, an expert on sustainable chemistry from the University of Birmingham, UK, said the study was ‘an impressive example of a fundamental advance in the understanding of mechanochemistry’.
‘While mechanochemistry … is readily conducted by, for example, ball-milling, extrusion or other types of agitation, the mechanistic understanding of such processes has remained at a very low level,’ he says. ‘As a result, the development of new and/or greener reactivity by milling mechanochemistry has so far remained largely based on trial-and-error.’
According to Friščić the study was an ‘ingenious application’ of theoretical modelling, atomic force microscopy and spectroscopy and represented a ‘major advance’ in understanding how directed mechanical force can affect the structure and reactivity of molecules on surfaces.
‘It provides an early glimpse into how mechanochemistry is able to provide access to unusual reaction products and selectivities,’ he says. ‘I am particularly fascinated by the [researchers] quantitatively establishing an important difference between how mechanical force, as uniaxial compression, will have a different effect on the reactivity of a surface-bound molecule compared to general hydrostatic pressure.’
‘Establishing how the proximity of a surface can modify reaction trajectories is a superb achievement,’ he adds. ‘While this particular report is not directed towards making chemical synthesis greener and cleaner per se, it is making a major contribution towards bringing mechanochemistry … one more, very important, step closer to rational understanding design.’
References
Y Zholdassov et al., Science, 2023. DOI: 10.1126/science.adf5273















1 Reader's comment