Silicon and ruthenium could form a quadruple bond, according to quantum calculations done by scientists in India. The hypothetical quadruple bond would be the first involving a heavier p-block element.1

Quadruple bonds have been reported between transition metals and elements in the first p-block row of the periodic table, such as carbon and boron. And last year, Ankur Guha’s group at Cotton University calculated that a quadruple bond between carbon and iron in CFe(CO)3 should be possible, but that SiFe(CO)3 would contain a triple bond.
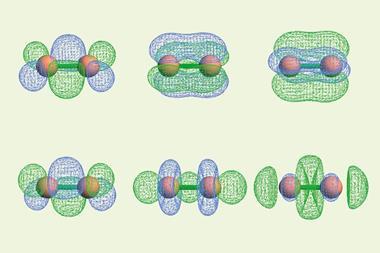
In this new study, Guha’s team decided to replace iron with ruthenium for a more energetically favourable configuration and analysed the bond structure of the molecule SiRu(CO)3 using computational techniques such as density functional theory. Their calculations revealed a quadruple bonding interaction between the silicon atom and ruthenium fragment.
Despite having four valence orbitals available for bonding, silicon usually fails to form quadruple bonds with transition metals, as longer bond lengths lead to unfavourable orbital configurations. SiRu(CO)3, however, can adopt C3V symmetry with a 1A1 singlet electronic state, causing effective orbital overlap between silicon and ruthenium.
Guha’s team suggest it is ‘quite likely’ that experimentalists will be able to make this molecule.
References
A J Kalita et al, Phys. Chem. Chem. Phys., 2021, DOI: 10.1039/d1cp00598g
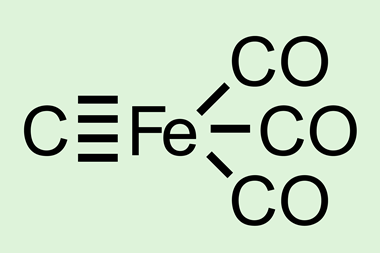






No comments yet