Researchers look to psychoactive substances for tips on drug development
Scientists in the UK have uncovered a surprising mechanism allowing some molecules to pass through the blood–brain barrier. This membrane is a source of frustration for chemists designing drugs, as its purpose is to block foreign substances from entering the brain. However, cocaine may lead researchers to a solution.
Current theories on how molecules pass through the barrier rely on defining motifs that must be included in the molecule. This often fails to account for the more complex conditions found in the body. ‘If you’re trying to design a drug that can treat brain related diseases, pharmaceutical companies have a massive problem,’ reflects Sylvia McLain, from the University of Oxford. ‘We’re looking at the fundamental physics behind how they actually interact with the blood–brain barrier so we can develop a bigger theory about how molecules do this.’
The work, carried out by Andrew Johnston, a DPhil student in McLain’s group, combines a number of techniques, including neutron diffraction, on solutions of cocaine, providing more biologically relevant information than the crystal structure alone.
‘This work is an extremely exciting application of neutron scattering,’ says Neal Skipper, a specialist in this technique at University College London in the UK. ‘These experiments illustrate the way that neutron and x-ray scattering can help us to understand how biological molecules function in solutions.’
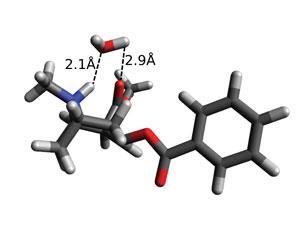
Up until now, it was though that cocaine had to be deprotonated to penetrate the blood–brain barrier. McLain and her team uncovered that conformation and hydration were far more significant. Rather surprisingly, a water mediated internal hydrogen bond appears to pull cocaine into a formation that shields its hydrophilic regions. In this state only the carbonyl oxygens and the amine group form directed hydrogen bonds with water, with the ester and benzene not forming any at all. This enables cocaine in solution to pass through the lipophilic environment of the blood–brain barrier without needing to be deprotonated as previously thought.
McLain emphasises the need for caution, as the mechanism has not yet been confirmed in the more complex environment of living organisms. What is clear from this work is how important it is to understand a molecule’s conformation in solution when designing drugs to reach the brain.
References
This article is free to access until 27 January 2016
A J Johnston et al, Phys. Chem. Chem. Phys., 2016, DOI: 10.1039/c5cp06090g












No comments yet