
A carbon dioxide-fixing enzyme with a modification unlike any other ever found in plants has been discovered. This modification has now been engineered into other plants and could help make photosynthesis in crop plants far more efficient, increasing yields and speeding growth.
Rubisco is an essential enzyme made of several subunits that work together to capture carbon dioxide, which plants then turn into sugars. However, it suffers from a tendency to react with oxygen, creating a toxic byproduct, 2-phosphoglycolate.
Many crop species use the photorespiration cycle to recycle this byproduct at a high energy cost while releasing carbon dioxide back into the atmosphere. This costs the plant in photosynthetic output and growth.
In algae, unique structures called pyrenoids have evolved to concentrate Rubisco and carbon dioxide in these organelles and reduce interactions with oxygen, making the process more efficient and reducing the synthesis of toxic byproducts. Algae create these pyrenoids by joining their Rubisco enzymes together using specific linker proteins. Reviews of photosynthetic efficiency across species estimate that pyrenoids increase carbon dioxide assimilation three-fold. Modelling estimates predict that introducing this carbon concentration mechanism into crops lacking one could increase efficiency by 30–60%.
‘Fascinatingly, across very distantly related algal lineages spanning over a billion years of evolution, different linker proteins have evolved independently but achieve the same result,’ says corresponding author Fay-Wei Li, a plant biologist at Cornell University.
Hornworts are the only land plants with pyrenoids and more closely related to crop species, meaning their pyrenoids are likely easier to introduce into crop plants than the distantly-related algal proteins. But attempts to find linker proteins in hornwort have been unsuccessful and the mechanism behind Rubisco concentration in these plants remained a mystery.
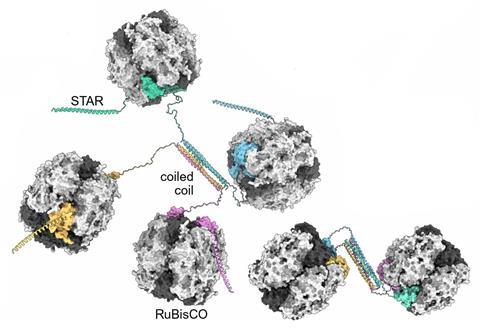
The first clue to what was behind this came when members of Li’s lab noticed an odd Rubisco isoform while designing tagging experiments in hornwort. ‘Immediately we suspected something interesting is going on,’ said Li.
Genome sequences revealed a 102 amino acid extension to the C-terminus of the Rubisco small subunit. They named the isoform RbcS-STAR and found that it formed coiled–coils, structures known to be involved in protein–protein interactions.
This represented a possible new mechanism for concentrating Rubisco that is different from algae. ‘In hornwort, it’s an extension of the small subunit and in other green algae, it’s basically an additional subunit that touches the small subunit,’ says Tobias Erb at the Max Planck Institute for Terrestrial Microbiology in Marburg, Germany.
Several experiments confirmed RbcS-STAR’s involvement in Rubisco concentration including mutations, genetic analyses and fluorescent tagging. ‘The critical functional test was expressing RbcS-STAR in Anthoceros fusiformis, a closely related hornwort that naturally lacks pyrenoids and watching pyrenoid-like structures form de novo,’ said Li.
After expressing it in Arabidopsis, a model system for many crop species, ‘it not only assembled into a functional Rubisco holoenzyme but also induced prominent electron-dense condensates in the chloroplast stroma’, Li said.
For Howard Griffiths, retired plant ecologist and emeritus fellow at Clare College at the University of Cambridge, the result is impressive. ‘I think there was a combination of both the hornworts being extremely clever, and then [the scientists] being extremely clever in finding out that mechanism and getting it to operate in higher plants,’ says Griffiths, who co-authored a perspective piece about the discovery. However, transplanting a functional pyrenoid into crop species like rice will require more than one Rubisco subtype. ‘You’ve got to have a system to deliver high CO2 to it,’ he says.
Li agrees. ‘We have built a Rubisco house, ie pyrenoids, but to actually functionalise it, we need to install the HVAC system to deliver CO2.’
Erb is fascinated by what the work reveals about biophysical strategies like condensing for improving reactions in other biotechnologies. Physically bringing enzymes closer together to increase efficiency, ‘it’s a very intriguing strategy’, he says. ‘If you can organise it in a condensed fashion, the substrate would effectively be within this condensate and move from A to B much quicker.’
References
T A Robison et al, Science, 2026, 391, 1070 (DOI: 10.1126/science.aea0150)












No comments yet