As fossil fuel usage is phased out to meet climate policy goals, an emerging challenge is discovering alternative synthesis routes for valuable chemicals manufactured using petrochemicals. With this in mind, researchers in China have genetically engineered Escherichia coli to generate the well-known painkiller paracetamol, starting from glucose or glycerol rather than benzene derivatives.
While the industrial synthesis of paracetamol has been carefully tuned to meet global demand, the process is far from ideal. ‘The substrates are all from the non-renewable petroleum derivative benzene, the intermediates are toxic and environmentally hazardous, [there are problems with] the safety [and] the unwanted ortho-isomer of benzene nitration, [and] a large amount of iron sludge and sewage will be produced,’ explains Wei Huang from the Qingdao Institute of Bioenergy and Bioprocess Technology.
Now, Huang and colleagues have designed a strain of bacteria that can make paracetamol through a biosynthetic pathway. E. coli can already produce the compound chorismic acid from glucose or glycerol through its shikimate pathway, and then metabolise chorismic acid to form p-aminobenzoic acid, albeit in low quantities. Paracetamol can be obtained from p-aminobenzoic acid in two biosynthetic steps – oxidative decarboxylation and regioselective acetylation – but E. coli does not have the required enzymes natively. By improving the existing pathway to p-aminobenzoic acid and then using genetic engineering to insert genes encoding enzymes from other species, Huang’s team extended the biosynthetic pathway all the way to paracetamol.
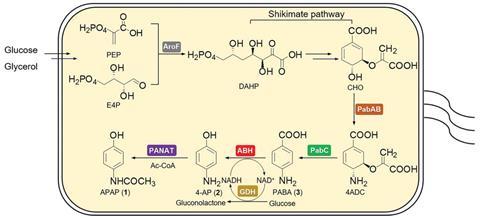
While its native biosynthetic pathways produce p-aminobenzoic acid, E. coli only form this in small quantities as the required biosynthetic enzymes are themselves only generated in small amounts. To solve this, the researchers inserted into E. coli additional copies of the corresponding genes, redesigned for increased expression to produce more of these enzymes. And as the next two steps needed enzymes that E. coli do not natively produce, the team then inserted new genes encoding these proteins, making use of biosynthetic pathways in edible button mushroom Agaricus bisporus and virulent pathogenic bacterium Pseudomonas aeruginosa.
‘You can take enzymes from across nature, from really odd locations, and combine them in one single cell, and I think this is a nice example of what you can do with that,’ comments synthetic biology expert Stephen Wallace from the University of Edinburgh, UK.
To further improve the biosynthetic yield, Huang‘s team used mutation studies to alter key residues in the newly added biosynthetic enzymes and selected mutants with superior turnover and selectivity. Optimising the fermentation conditions led to paracetamol yields of 942.5mg per litre of glucose-fed culture on a five-litre scale. Alternatively, using a shorter biosynthetic pathway saw 95% of p-aminobenzoic acid feedstock converted and a yield of 4.2g paracetamol per litre of culture in nine hours. ‘Our strategy makes it possible for bacteria to produce this traditional drug as cell factories,’ says Huang.
Although this work is the first example of a prokaryote synthesising paracetamol in usable quantities, Huang says scaling up the method will require further development: ‘The entire designed biosynthetic pathway needs to be further optimised. The fermentation and reaction conditions also need to be improved.’ Wallace agrees: ‘Multigrams per litre is approaching something that can be industrially scaled up. The trouble with paracetamol is that it’s made on such a vast scale, and the prices of petrochemical feedstocks are so low, that this probably isn’t going to be economically viable right now. But it’s precisely the research that we need to be doing in light of diminishing fossil fuels.’
References
F Hou, M Xian and W Huang, Green Chem., 2021, 23, 8280 (DOI: 10.1039/d1gc02591k)










No comments yet