Mechanochemical route to naphthalenediimide radical ions
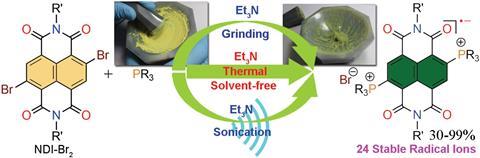
Researchers in India have devised the first eco-friendly method for generating organic radical ions.1
Organic radical ions, familiar to many chemists as reactive intermediate species, have gained interest over the past century as their tuneable molecular magnetism could benefit data storage materials and MRI probes. However, in stark contrast to the green progress made in many areas of organic chemistry, until now, not a single example of a green or environmentally friendly method for making organic radical ion existed.
Pritam Mukhopadhyay, of Jawaharlal Nehru University, and his team have spent years investigating radical ion design. ‘It’s a challenging and highly rewarding area of chemistry given its inherently reactive nature and instability,’ he tells Chemistry World. The hypersensitivity of these species meant few had even considered a synthesis using green methods. A typical radical ion synthesis requires hazardous reducing or oxidising agents, to essentially rip molecules apart and force these ions into existence. Even then, stabilising the resulting species necessitated some pretty nasty solvents.
Spurred on by their recent unprecedented discovery of a stable set of naphthalenediimide radial ions,2 Mukhopadhyay decided to try a greener route. The team reimagined the synthesis with sustainability in mind: they removed all solvents, screened non-toxic reactants, and found a set of conditions that with applying a bit of heat, worked, and with a decent yield. Better yet, the team found that instead of heat, sonication or even just a bit of hard mortar and pestle grinding gave the reactants just enough energy to push the components into forming radical ions.
The discovery opened the flood-gates for Mukhopadhyay’s team, who report not one, but 24 structurally diverse, ambient stable radical ions, all synthsised and isolated with good yields using cheap and environmentally friendly methods.
References
1 S Kumar and P Mukhopadhyay, Green Chem., 2018, DOI: 10.1039/c8gc01614c
2 S Kumar et al, J. Am. Chem. Soc., 2014, 136, 12004 (DOI: 10.1021/ja504903j)










No comments yet