Scientists in Germany have measured a zeptosecond time delay between a photon hitting a dihydrogen molecule and its electrons being ejected. They say this ‘birth-time delay’ corresponds to the time taken for the photon to travel the length of the molecule.
Photoionisation has been used for decades to explore the electronic structure of atoms and molecules. By measuring the energy of electrons ejected when a photon is absorbed, and calculating the difference in energy between the incident photon and the ejected electron, scientists can map the binding energies of electrons within materials.
These days, with modern equipment capable of attosecond time resolution, researchers can also glean useful information from measuring the time delay between the photon incidence and the electron emission. The Wigner delay, which is typically tens of attoseconds, results from complex interactions between the phase of the atomic or molecular orbitals in different parts of the molecule and the phase of the incident photon. ‘The theory on that started something like 60 years ago but it’s just very recently that they’ve been able to address things like this in experiments,’ says Till Jahnke of Goethe University in Frankfurt, Germany.
Now, Jahnke and Goethe University colleagues led by Reinhard Dörner have taken on an even tougher challenge: measuring the minuscule effects arising from the time it takes for light to travel across a molecule. Since the speed of light is finite and constant, they reasoned, one side of a molecule should feel the effect of an incident photon a tiny fraction of a second sooner than the other. To test this, they used an ingenious but well-established version of the double slit experiment used to demonstrate the wave nature of light in which the slits are replaced by the two atoms of a diatomic molecule.
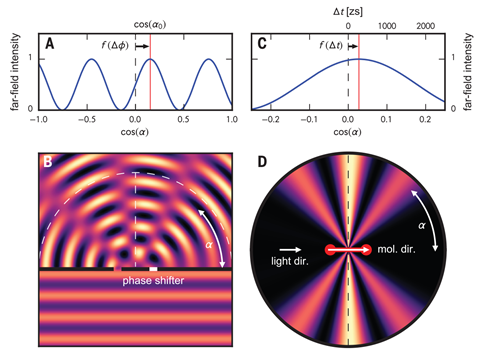
When the researchers fired x-ray photons at H2 molecules, each bond initially released a high-energy electron from one of the two atoms with equal likelihood. The researchers then reconstructed the orientation of each molecule at the point of ionisation to find out which hydrogen atom had been closer to the incident photon, before calculating a diffraction pattern of emitted electrons relative to the axes of the bonds. If the incident photon’s direction had been irrelevant, the diffraction pattern should have been symmetrical. Instead, the researchers detected a significant shift in the fringes, showing that electron excitation occurred first at the part of the molecular orbital nearest to the photon, rather than across the whole orbital simultaneously. The other side of the molecular orbital then experienced the photon’s potential slightly later, in this case just 247 zeptoseconds (1 zs = 10-21 s), leading to a phase difference between two interfering electron waves that results in a shift in the diffraction pattern.
David Villeneuve of the University of Ottawa in Canada is impressed: ‘I think this is a heroic experiment, where tiny shifts in the direction of the electron can be measured,’ he says. ‘The shifts are interpreted as a speed-of-light delay across the tiny molecule. Who would have thought that that was important?’
Hrvoje Petek of University of Pittsburgh in Pennysylvania, US, however, cautions he does not think it ‘provides new knowledge on anything that has some useful physical significance’.
Still, the researchers believe it should be a general phenomenon and are setting out to look for the effect in other systems: ‘The next step for me will be to look at some more complicated molecules such as N2 or carbon monoxide,’ says team member Sven Grundmann.
References
S Grundmann et al, Science, 2020, 370, 339 (DOI: 10.1126/science.abb9318)












No comments yet