New measurements have revealed rising levels of five potent ozone-depleting gases, despite rigorous international controls over their production. A cross-disciplinary team reported a 2.6-fold increase in the emissions of five regulated chlorofluorocarbons (CFCs) between 2010 and 2020, which could undermine the progress of current environmental targets.
CFCs are the fully-halogenated derivatives of simple hydrocarbons and their use was formerly widespread as refrigerants and aerosol propellants. Their devastating environmental impact, most notably causing the hole in the ozone layer, led to tight global regulation under the Montreal Protocol.
‘CFCs are chemically inert in the lowest level of the atmosphere, but in the stratosphere they are broken down through photochemical reactions,’ explains James Keeble, an atmospheric science researcher at the University of Cambridge UK. ‘The chlorine that is released then destroys stratospheric ozone through catalytic reactions.’

But while ozone damage has been the major focus of regulation, CFCs are also potent greenhouse gases with global warming potentials thousands of times greater than carbon dioxide. ‘In terms of climate impact, the emissions of these five CFCs in 2020 were around the same as the [total] CO2 emissions from Switzerland that year,’ says study leader Luke Western from the University of Bristol, UK.
In real terms, these recent CFC increases are relatively small but they raise questions about whether more comprehensive regulation is necessary. ‘The Montreal Protocol has been very successful in reducing the production of CFCs for emissive use,’ says Brad Hall, an atmospheric science researcher at the NOAA Global Monitoring Laboratory, US. ‘But there are exceptions for chemicals used as feedstocks and process agents. Exemptions were granted to allow continued production of substances allowed under the protocol such as HFCs.’
Hydrofluorocarbons (HFCs) are the partially halogenated cousins of CFCs, which crucially do not contain ozone-depleting chlorine. Their otherwise similar properties made them ideal replacements following the ban and HFC-125 and 134a (CHF2CF3 and CF3CH2F, respectively) became the coolant of choice for refrigeration and air conditioning units. However, of the five CFCs identified by Western’s team, three are feedstocks, intermediates or byproducts released during the preparation of these alternatives. Successive halogen exchange reactions remove the problematic chlorine atoms but up to 8% of the material may end up as CFC-113a (CF3CCl3), 114a (CF3CCl2F) and 115 (CF3CClF2). After comparing estimates of HFC production and leakage with the measured emissions rates, the team concluded that a significant proportion of these increases likely arise from processes permitted under the Montreal Protocol.
However, the two other CFCs measured by Western’s team, CFC-13 (CClF3) and 112a (CClF2CCl3) have no current uses to explain these increases. ‘CFC-13 can be produced during plasma arc destruction of CFC-12 and from aluminium smelters, but it seems unlikely that this would be driving the change,’ comments Western. ‘CFC-13 can be a byproduct during the production of CFC-11 and 12 and we know there was unreported production of these chemicals in east Asia. But this appears to have stopped, whereas emissions of CFC-13 have continued to increase.’
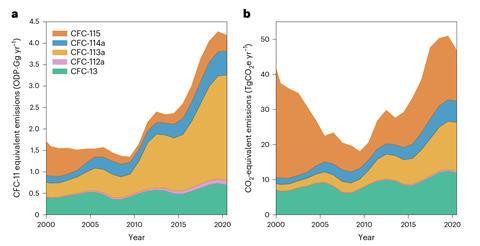
At present, these emissions have had a relatively small impact on the atmosphere and the ozone layer remains on track for a full recovery within the next 50 years. However, researchers across the field are keen to monitor these changes. ‘If the sources are not fully identified and the emissions continue to increase, the impact will be more significant,’ says Keeble. ‘International efforts to identify whether CFC emissions are decreasing as expected are vital to ensure the continued success of the Montreal Protocol and ozone recovery.’
References
L M Western et al, Nat. Geosci., 2023, DOI: 10.1038/s41561-023-01147-w

















No comments yet