Researchers on opposite sides of the Atlantic have unveiled new findings on the ways two different types of scattering resonances affect the interactions of ultracold molecules. The papers mark a further step towards coherent control of chemical reactions and could potentially aid the creation and understanding of exotic quantum matter.
Near absolute zero, the thermal motion that blurs the quantum behaviour of atoms and molecules at room temperature almost ceases. Researchers therefore seek to understand chemistry at the quantum level and potentially go on to manipulate it.
In 1998, Wolfgang Ketterle at the Massachusetts Institute of Technology (MIT) and colleagues made the first observations of ‘Feshbach resonances’ between trapped sodium atoms in magnetic fields.1 The atoms were normally non-interacting, so collisions were elastic and the sample remained trapped. However, at a specific field strength, two electronic energy states had the same energy. This allowed for inelastic collisions as the quantum wavefunctions of the particles interfered, forming unstable intermediate states. This disrupted the sample and caused it to be destroyed almost immediately. Molecules are much trickier as they have many more internal degrees of freedom, but in 2022 Ketterle’s group tailored the quantum wavefunctions of colliding sodium atoms and sodium lithium molecules.2 After searching a range of magnetic fields, they found two Feshbach resonances at which the loss rate of the sample increased, in one case more than a hundredfold.
In the first of the new papers, the MIT researchers went one stage further and studied collisions between two sodium–lithium molecules.3 They expected to see nothing. ‘If you compare molecule–molecule collisions with atom–molecule collisions, the number of possible states can be a few orders of magnitude higher, so the resonant features could be smeared out,’ says Ketterle’s PhD student Juliana Park, the work’s lead author. ‘But the theories are not perfect so I checked it.’
She observed a single, extremely narrow peak at which the loss rate jumped more than two orders of magnitude. The researchers propose a model in which, at this specific field, waves entering the Feshbach resonance interfere destructively with waves leaving it, preserving the intermediate and causing more loss. ‘We’re not sure if this is the right model,’ says Park. ‘If other theorists can explain it better, we’ll be happy to know more.’
Experimental confirmation
Useful as Feshbach resonances have proved to Ketterle’s group and numerous others, they have limitations. Notably, they exist only in molecules with magnetically tunable electron energy levels, which requires them to have unpaired electrons. In 2003, however, theoretical physicist John Bohn at University of Colorado Boulder in the US predicted that, if a microwave-frequency oscillating electromagnetic field were applied to any polar molecule, the molecular dipoles could interact through ‘field-linked resonances’.4 In the second of the new papers, scientists in Germany have experimentally confirmed this.5
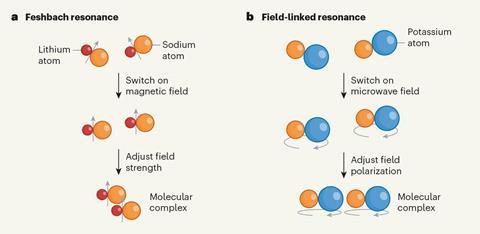
The resonances occur because the applied field modifies the interaction potentials between molecules such that they become attractive at long distances but repulsive closer up, creating intermolecular potential wells that bind pairs of molecules into ‘supermolecules’. ‘This is a kind of long-range bound state,’ explains group leader Xin-Yu Luo at the Max Planck Institute of Quantum Optics. ‘There is a long-sought so-called p-wave superfluid that was first discovered in helium-3 in the 1970s,’ Luo says. ‘Here, in principle, we should be able to create a p-wave superfluid from polar molecules, but with a highly controllable interaction because the shape and symmetry of the intermolecular potential can be flexibly tuned by the polarisation, frequency and power of the microwave.’
David DeMille of the University of Chicago says both papers are remarkable. The MIT work underlines that ‘what theorists predict isn’t necessarily true’, he says. ‘It was really widely understood that these Feshbach resonances, which are so widely used in atoms, probably couldn’t be seen in molecules.’ This reinforces the importance of the other work, he says. ‘In the resonance the Max Planck group sees, the molecules are quite far apart. They do a sort of dance where they never get close together and yet they’re bound to each other purely by their electric dipole interactions… It’s really quite unlike any molecular state that’s ever been seen before. For others in the field like me that’s pretty cool, but what’s really exciting is that you can use these states to influence how the molecules are interacting in a controlled way.’
References
1 S Inouye et al, Nature, 1998, 392, 151 (DOI: 10.1038/32354)
2 H Son et al, Science, 2022, 375, 1006 (DOI: 10.1126/science.abl7257)
3 JJ Park et al, Nature, 2023, 614, 54 (DOI: 10.1038/s41586-022-05635-8)
4 AV Avdeenkov and JL Bohn, Phys. Rev. Lett., 2003, 90, 043006 (DOI: 10.1103/PhysRevLett.90.043006)
5 XY Chen et al, Nature, 2023, 614, 59 (DOI: 10.1038/s41586-022-05651-8)
















No comments yet