Meera Senthilingam
This week, a brief history of crowd control, with Simon Cotton
Simon Cotton
The use of chemicals in crowd control goes back a long way. There are accounts going back hundreds of year of Chinese and Japanese use of powdered pepper as defensive agents. The active ingredient in modern pepper sprays is capsaicin, the molecule that gives chilli peppers their bite – if you’ve been chopping up hot chillies, capsaicin is the reason you should wash your hands before you go to the toilet as well as afterwards.

Most chemicals used in riot control are man-made, however. For many years, CN was used to incapacitate people; if you select four letters from the chemical name, methyl chloroacetophenone you get Mace, the name by which it is often known. Mace is thought to be a carcinogen and also may have caused a few fatalities in its use, so the much safer CS has supplanted it since the 1960s.
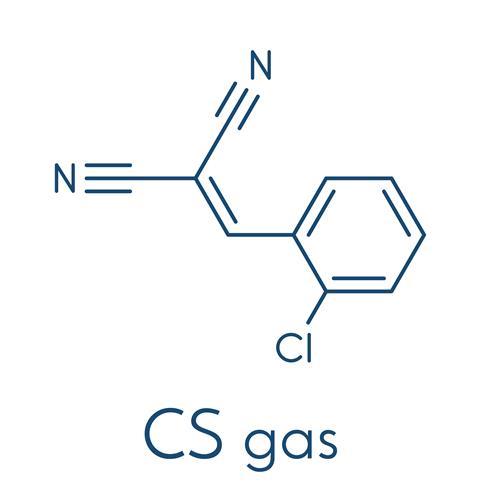
The first thing you need to know about CS is that it isn’t a gas. CS is a convenient shorthand name for the molecule 2-chlorobenzalmalononitrile, which has a melting point of 93°C, so when it is used as a riot control agent, it is usually dissolved in a solvent like methyl isobutyl ketone.
The chemical that we know as CS was first reported in 1928. It wasn’t the product of chemical industry or even a top university – it was discovered by two chemists at an American liberal arts college, set in a picturesque part of the state of Vermont. Ben Corson and Roger Stoughton, of Middlebury College, were systematically investigating the reaction between carbonyl compounds, either aldehydes or ketones, and malononitrile, a methane molecule in which two hydrogens have been replaced by cyanide groups. They synthesised several new compounds. CS was one of these. Deep into their 13-page report we read ‘certain of these dinitriles have the effect of sneeze and tear gases. They are harmless when wet but to handle the dry powder is disastrous.’
No one was much interested in these compounds at the time, but someone must have read the paper because towards the end of the 1950s researchers at Porton Down, the British chemical defence unit, pioneered its use, and soon it was in use in several countries. The incapacitating agents like CN and CS are usually referred to by two letters, the first being C, though it may be no coincidence that C and S are the initials of Corson and Stoughton, its joint discoverers.
CS is reported to be safe, as far as such agents go
During the Vietnam war, from the mid-1960s into the early 1970s, American forces used CS as a means of clearing enemy troops from underground bunkers, whilst British troops in Northern Ireland used CS from 1969 into the early 1970s to control riots. Although it is now prohibited from use in war by the Chemical Weapons Convention of 1993, its use by police forces is generally legal. CS has been widely employed in many parts of the world for riot control, in places like Bahrain, Nepal, South Korea and Egypt.
CS works in much the same way as other eye irritants, whether it is formaldehyde from wood smoke and cigarette smoke, or syn-propanethial S-oxide, the compound released when you peel onions. These stimulate the mucous membrane of the eye to act protectively; the irritation is followed by a nerve message back to the lachrymal glands, which respond by washing the irritant away with tears.
With a lethal dose some 50,000 times the effective dose, CS is reported to be safe, as far as such agents go. If it were to be used in confined spaces, then concentrations can rise to lethal amounts, as well as causing panic. A number of fatalities have arisen accompanying its deployment, one example being the siege of the Branch Davidian complex near Waco, Texas, on 19 April 1993. A toxicologist quoted in the subsequent Danforth report explained that if people exposed to high levels of CS are not able to leave a room, ‘there is a distinct possibility that this kind of CS exposure can significantly contribute to or even cause lethal effects’. Most recently CS gas appears to have been used to suppress peaceful protest in the confrontation between police and protestors in Ferguson, Missouri.
The dividing line between a dispersing agent and a chemical weapon seems very narrow at times.
Meera Senthilingam
Birmingham University’s Simon Cotton there, with the controlling chemistry of CS gas and its consequences. Next week, Brian Clegg is mystified.
Brian Clegg
When I read about them in my youth, there seemed something magical in the description of the acetylene or carbide lamps that were used on early motor vehicles. The idea that adding water to the lamp started a process that could generate a flame seemed wonderfully counter-intuitive.
Meera Senthilingam
The chemisty behind this seeming contradiction is explained in next week’s Chemistry in its Element. Until then, thank you for listening, I’m Meera Senthilingam













No comments yet