Chemists are working with toxicologists at an earlier stage to avoid problems further down the chain, as Emma Davies reports
A growing number of chemists are turning to toxicological tools to screen for potential environmental and health hazards during the early stages of chemical design. Their work comes under the fourth of the 12 principles of green chemistry: designing safer chemicals. This principle is the ‘most scientifically exciting, the most practically applicable for industry and the most urgent for human health and the environment’, says Paul Anastas. He is director of Yale University’s Center for Green Chemistry and Green Engineering and came up with the principles in 1998 together with John Warner, then at the University of Massachusetts in the US.
Last year, Anastas’ brother Nick, at the US Environmental Protection Agency (EPA), coined the phrase ‘green toxicology’ to bridge the gap between toxicology and sustainable molecular design.1 ‘It focuses on a particular application of the principles of toxicology to address certain problems, in this case, articulating design rules that can be used by synthetic chemistry to design products and reduce hazard,’ he explains.
Nick Anastas leads the EPA’s New England Green Chemistry Challenge, which aims to increase the adoption of those 12 principles across the region, with the ultimate goal of a sustainable economy. In December 2012, the EPA in New England teamed up with the Johns Hopkins Centre for Alternative Animal Testing (CAAT) to run a toxicology and sustainable molecular design conference and a green toxicology seminar is scheduled for November 2013.
Testing times
‘Green toxicology is similar to the idea in the pharmaceutical industry of frontloading toxicity assessments – fail early, fail cheaply,’ explains CAAT director Thomas Hartung. ‘While expensive and long animal tests are of little help, in vitro and in silico approaches promise to do the trick.’ At such an early stage in a chemical’s development, false negative and false positive results are considered acceptable. ‘Unlike their regulatory use, the applications do not need the ultimate proof of predicting each and every possible adverse effect as the regulatory evaluation is coming later anyway,’ says Hartung.
Both groups benefit, he adds. ‘Chemists don’t like to see substances die late on and toxicologists have found new customers who don’t require 10 years of validation before they use their tools.’
Toxicology has traditionally been taught more from a biology perspective than from a chemical structure one
Marty Mulvihill
Marty Mulvihill, director of the Berkeley Center for Green Chemistry, US, agrees. ‘The toxicologists that I work with are excited to think about toxicology from a design perspective, to think about early intervention and to think more about chemical structures.’
Toxicologists tend to test chemicals already on the market, but Mulvihill says that working alongside chemists designing new molecules can also provide insights into where the next area of interest is. Together with a toxicologist colleague, Mulvihill runs a course on toxicology for green molecular design. ‘Toxicology has traditionally been taught more from a biology perspective than from a chemical structure perspective. That has been one of the big hurdles – why there has been so little toxicology taught during chemistry degrees,’ he says.
‘As chemists we are taught structure–function relationships; that’s a big part of how chemists think. What we need to do is draw structural analogies for problems like degradation in the environment, or potential to bind to hormone receptors,’ says Mulvihill.
Mulvihill considers industry to be a step ahead of academia when it comes to recognising the need for early-stage toxicology screening. ‘The public are already asking for it and regulators are considering it but academic journals aren’t yet looking for it,’ he says. ‘At the moment, I think the drivers in the real world are more pressing than the drivers in academia.’
Industrial embrace
As a case in point, at German chemical company BASF, toxicologists work very closely with synthetic chemists to eliminate lead molecules with potential hazards.

Bennard van Ravenzwaay heads BASF’s experimental toxicology and ecology unit, which uses a suite of tiered screening tests for emerging chemicals, starting with in silico and in vitro tests. For van Ravenzwaay, a key advantage of the screening is the fact that it makes the testing regime far more efficient. When he started his job 14 years ago, he was struck by the vast quantities of data produced by toxicity testing. ‘Out of 40 toxicology studies we would maybe use five or six. The trouble was that the scientists had no way of knowing which of the studies would be relevant for a new molecule. One of the drivers for me personally was to make the system more efficient,’ he says.
Importantly, the suite of screening tests has dramatically reduced the quantity of a substance that the chemists designing a new molecule need to produce. ‘If you have a 15-step process, synthesising 200g for the toxicologists takes weeks and weeks of work and if you need to do that for a whole series of compounds, because you’re in a selection phase, it’s absolutely impossible,’ says van Ravenzwaay. ‘So green toxicology is a beautiful enabler because we don’t need 200g, we can use 100mg.’
BASF spends about €2 million (£1.7 million) per year developing alternatives to animal tests. A current focus is on tests to predict developmental toxicity. ‘That’s pretty difficult because scientifically it’s not well-known what type of mechanisms cause the effects,’ says van Ravenzwaay.
‘What we have seen recently [in the past two to three years] is a paradigm shift in chemical safety testing,’ says Mike Witt, global director of toxicology and environmental research and consulting at Dow Chemical Company in the US. His unit provides environmental and toxicological testing services to all Dow businesses. ‘What we are doing is applying new tools that factor safety into design and selection. We find ourselves getting involved earlier in the process of product selection, specifically molecular design. By integrating ourselves early in the process we work very closely with those in R&D to look at the structures of molecules,’ he says.
Witt uses the term predictive toxicology to describe Dow’s ‘integrated suite of assessment tools’, many of which use quantitative structure–activity relationships. The collection of tools speeds up the evaluation of molecules and reduces the use of lab animals, yet brings the challenge of dealing with the truly vast amounts of data produced by high-throughput techniques, he says.
Fishy business
As well as its in silico techniques, Dow also makes use of early-stage cell tests and tests on zebrafish. ‘The tests are highly predictive of what would happen in a mammalian system and we can do them very rapidly and cheaply,’ says Robert Tanguay from Oregon State University, US.

‘Chemists can give their lead materials to us and in a matter of days we can give some comparative results. Lowering the barriers of doing whole animal testing has really helped people to take a look at chemicals early on,’ he says.
The set-up is highly automated and the lab prepares 80,000 zebrafish eggs per day for tests. Automated instruments remove the protective shell from each egg to increase the sensitivity to chemical exposure. Further down the line, robots gently place the zebrafish embryos in individual wells of water. Each embryo is then monitored by more instruments to see if chemical exposure affects development.
Tanguay has done a great deal of nanotechnology toxicity testing.2 Much of the work looks at surface effects. ‘Primarily, the surface is what drives the biocompatibility,’ he says. ‘We are able to change the conditions to alter agglomeration.’ Key to such studies was the discovery that the zebrafish embryos can survive in reverse-osmosis water, with all of the ions removed.
Homing in on endocrine disrupters

Tanguay recently worked with Terry Collins, director of the Institute for Green Science at Carnegie Mellon University, US, to test the toxicity of a range of catalysts. Collins is a green chemist – ‘one of the originals’, he says – best known for designing small molecules that mimic oxidising enzymes.
Collins partitions chemistry into six major challenges, which he pictures as book shelves. ‘My top shelf is endocrine disruption. We can’t be subjecting living things to chemicals that produce major biological changes,’ he says.
His catalysts, called TAML peroxide activators, work at ‘unbelievably small concentrations’ in water and can remove endocrine disrupters through oxidation. They are on the road to commercial success, thanks to the discovery that they will remove the problem compound ethinyl oestradiol (EE2), used in contraceptive pills, from water.
‘We are in a remarkable position to come in and attack endocrine disruption in water,’ says Collins. He has recently teamed with endocrine expert Susan Jobling from Brunel University, UK, and plans are afoot for a UK water company to test a TAML catalyst in a pilot plant in Exeter. All that’s needed is a pump for the catalyst and another for peroxide. ‘We’re very excited: we are on the move to bring this stuff into the real world,’ says Collins.
Yet for many years, Collins was worried about the potential toxicity of his catalysts. ‘It bothered me immensely that I could not assert to anybody that my catalysts were any safer than EE2,’ he recalls.
Collins was one of an expert team of environmental health scientists and green chemists to come together to ‘design endocrine disruption out of the next generation of chemicals’. They took themselves away for a couple of retreats in California and the end result was the Tiered Protocol for Endocrine Disruption (TiPED),3 which describes five levels of toxicity testing: in silico; high-throughput in vitro and in vivo screens; in vitro whole cell activity assessment; fish and amphibian whole animal assessment; and mammalian whole animal assessment (see Stopping endocrine disruptors in their tracks).
Laura Vandenberg, who recently took up a new post at the University of Massachusetts in Amherst, US, was one of the TiPED team. ‘The basic idea is that if you are a chemist and you are going to design a chemical from scratch then we offer you tools in order to determine whether the new chemical has endocrine-disrupting hazard while also screening for other things such as general toxicity,’ she explains.
The screening tools are those used by environmental health scientists to test the toxicity of chemicals that are already on the market. ‘We tried to flip them on their head to determine whether a chemical has toxicity before it becomes an actual product,’ says Vandenberg.
‘These assays are not designed for regulatory actions, they are designed for green chemists who believe in making chemicals that are as safe as possible,’ she adds. TiPED is described as a ‘living protocol’ that evolves as assays are refined and new ones developed.
Collins has used TiPED to test TAML catalysts, with different groups taking responsibility for different testing tiers. For example, Bruce Blumberg, from the University of California, Irvine, tested the catalysts using oestrogen and androgen receptors. Meanwhile, Collins worked with Cheryl Watson from the University of Texas Medical Branch on tier three, while tier four testing was done on zebrafish with Tanguay.
The catalysts are typically used at concentrations of 100 nanomolar or less. ‘All of the catalysts came up clean at the lowest concentrations but three [out of seven] of the catalysts start showing developmental effects at 100 micromolar and 250 micromolar,’ he says. As a result, Collins will not develop those three catalysts for commercial use.
Ionic liquids
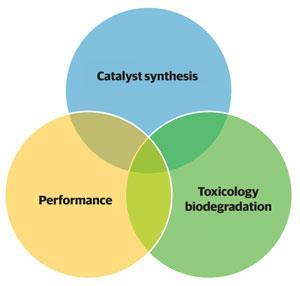
Nick Gathergood from Dublin City University, Ireland, also takes a green approach in his work on performing catalysis in ionic liquids. His team not only designs the ionic liquids to have low antimicrobial toxicity but also carries out tandem toxicity screening to direct the research towards ‘greener’ chemicals.
‘Our approach is to give equal consideration to performance, toxicology and biodegradation. The core ideal behind this is that the toxicity and biodegradation of a chemical is independent of the application or performance,’ explains Gathergood.
A recent study of his found one of a range of novel ionic liquids to have high antimicrobial toxicity.4 ‘We suggest avoiding its use, especially as a solvent. If another research group is looking to use the same class of ionic liquids that we prepared in the paper, I would hope they would try the “safer” examples first, or even avoid trying the toxic one,’ he says. ‘Had we published the paper without the toxicity data, as almost every other catalysis group does, another group could unwittingly select the toxic ionic liquid instead of a safer one.’
Gathergood has worked with a group of international researchers to develop a ‘traffic light’ classification of performance, eco-toxicity and efficient synthesis to ‘direct our catalyst project towards less toxic catalysts’.5
Incorporating the toxicity testing into research routines has been straightforward, says Gathergood, with the iterative process of design, synthesis and evaluation of biological activity mimicking the basic concept of drug development.
He does not advocate extensive toxicological testing of every new compound, since this would ‘stifle productivity’, but recommends selective toxicological screening. ‘If we can convince chemists of the benefits of routine basic toxicology screening, that would be a major achievement,’ he says.
Touting toxicology
Berkeley’s Mulvihill also cautions against chemists taking on too much. ‘What I can do as a chemist is to teach people how to do that initial in silico screen – the first step in the process. It helps to build those bridges, helps to get the dialogue going so that we can look at things in greater depth but it can’t be just the chemist’s responsibility to do everything.’ All Mulvihill asks is that chemists ‘look a little bit deeper and apply their already-existing chemical knowledge in new and productive ways’.
It’s not about turning chemists into toxicologists – or vice versa
Paul Anastas
‘This is a tremendous scientific challenge,’ says Paul Anastas. ‘It’s a wonderfully rewarding area. What we’ve seen in recent years is not that we have solved the problem – far from it. What we’ve shown is that it’s possible.’
Green toxicology is a complementary approach, he adds. ‘It’s not about turning toxicologists into chemists or chemists into toxicologists. It’s giving each the insight and the awareness to inform the solution.’
Emma Davies is a science writer based in Bishop’s Stortford, UK
References
1 N Anastas in: Green Techniques for Organic Synthesis and Medicinal Chemistry, Wiley, 2012
2 Ki-Tae Kim and Robert L. Tanguay, Green Chem., 2013, 15, 872 (DOI: 10.1039/c3gc36806h)
3 T. T. Schug et al, Green Chem., 2013, 15, 181 (DOI: 10.1039/c2gc35055f)
4 R Gore et al, Green Chem., 2013, DOI: 10.1039/c3gc40875b
5 R G Gore et al, Green Chem., 2013, DOI: 10.1039/c3gc40992a













No comments yet