Mike Sutton looks at what we’ve learned about the moon’s chemistry in the 50 years since Apollo 11
On 20 July 1969, Apollo 11’s lunar module landed on the surface of the moon. A few hours later, on 21 July, Neil Armstrong and Buzz Aldrin stepped out. The mission was the culmination of the decade long Apollo programme to take humans to the moon. It was undoubtedly one of the crowning scientific and engineering achievements of the 20th century – but what did we learn about the moon?
During Armstrong and Aldrin’s two and a quarter hours spent outside the module on the moon, they collected around 20kg of rocks and other lunar material. The subsequent five Apollo missions that reached the moon also brought back samples, over 300kg in total.
When the Apollo astronauts returned with their treasures, chemists could begin investigating our satellite’s composition directly. This research still continues, as new analytical techniques are developed and fresh theoretical questions posed.
All at sea
When Galileo Galilei published his revolutionary telescopic observations in 1610, he attributed the changing patterns of light he saw on the moon’s surface to mountains catching the lunar dawn before it reached the lowlands. This conclusion was immediately challenged by academic philosophers, who regarded it as incompatible with Aristotle’s doctrine that all celestial bodies were perfect spheres.
Spectroscopy could detect elements in the sun, but it revealed little about the moon, which merely reflects solar light
By the late 17th century, however, it was widely accepted that the moon was a material body with topographical features resembling the Earth’s. Its darker and smoother areas were therefore called maria (seas), and its brighter and rougher regions terrae (lands). Its numerous craters were assumed to be volcanoes. In his 1687 Principia Mathematica Isaac Newton estimated the moon’s gravitational pull from measurements of the rise and fall of tides, and calculated its density to be eleven-ninths of the Earth’s – almost double the correct value.
Although 19th century physicists could measure the moon’s mass far more accurately, its composition remained mysterious. Spectroscopy could detect elements in the sun and stars, but initially it revealed little about the moon, which merely reflects solar light, minus the frequencies it absorbs.
In the 20th century, bigger telescopes and better photography produced more detailed maps of the moon’s surface. However, the relative importance of volcanism and bombardment in shaping that surface was vigorously disputed – notably in heated exchanges between Nobel prize-winning American chemist Harold Urey and distinguished Dutch–American astronomer Gerard Kuiper. Meanwhile US astronomer Ralph Baldwin’s calculations – indicating that most lunar craters were too big to be volcanic – were largely ignored until the space race started.
First landings

In 1959 the USSR’s Luna 2 probe hit the moon. Before the impact its instruments detected no magnetic field, implying the absence of an Earth-like core of liquid iron. Seismological observations later indicated that the moon’s iron core is very small, and mostly solid. When Luna 10 orbited the moon in 1966, its gamma-ray spectrometer analysed the radiation emitted from atomic nuclei that had been struck by high-energy cosmic rays. It detected elements indicating the presence of basalt – the most common terrestrial volcanic rock – in the darker regions still called maria. Astronomers had long ago concluded that these ‘seas’ contained no water, but the name survived.
In 1967 the US’s Surveyor 5 landed on Mare Tranquilitatis – the ‘Sea of Tranquillity’. It used alpha-particle back-scattering from elements on the surface to identify a specific type of basalt, commonly found in Greenland (basalt is not a single compound, but a mixture of different silicates in variable proportions). The next step was for astronauts to collect lunar samples.
Between 1969 and 1972 the US’s six Apollo landings yielded approximately 381kg of material, while three of the USSR’s robotic probes returned 0.326kg between 1970 and 1976. Scientists examined these specimens intensively, as it seemed unlikely that more would become available for many years. In 1982, however, another source was identified by New Zealand geochemist Brian Mason, the curator of meteorites at the Smithsonian Institute in Washington DC.

When Nasa sent him an unusual specimen from Antarctica, Mason quickly recognised its resemblance to material collected by astronauts. Its distinctive iron–manganese ratio later confirmed that it came from the moon – blasted free by some gigantic impact, and subsequently captured by the Earth’s gravity. More meteorites – over 190kg in total – have since yielded chemical and mineralogical evidence of their lunar origin. These bodies also contain radioisotopes generated by exposure to cosmic rays on the moon or in space. Inside Earth’s atmosphere, which muffles cosmic radiation, the formation of these ‘cosmogenic nuclides’ ceases, and as they have different decay rates their relative proportions indicate when a meteorite reached Earth.
Not cheese after all
Interpreting all this data is a problematic exercise, because the lunar surface is a composite entity. Unlike the Earth, the moon has no magnetosphere to prevent the solar wind depositing hydrogen (and lesser amounts of heavier elements) on it, and while the low lunar gravity allows much of this material to dissipate into space, more keeps arriving. The moon is also littered with meteors that differ significantly from its original surface material – which has itself been radically transformed by violent impacts.

Much of the lunar surface is covered with regolith, a powdery mixture of rocks that have been smashed and then repeatedly stirred by bombardment – a process selenologists call ‘gardening’. It often contains tiny glassy spherules, most of them probably created when meteoric impacts melted silicate rocks and scattered the resulting droplets, though some may be of volcanic origin. Bigger impacts released enough energy to fuse heterogenous mineral deposits into composite rocks – known as breccia – which subsequent bombardments shattered. Gradually, this chaotic record has been deciphered.
Before the space age began, we knew the moon’s density was about 60% that of the Earth’s. Data from orbiting instruments and surface samples confirmed the predominance of lighter elements in its crust. Chief among these are oxygen (45% by weight) and silicon (21% by weight), mostly combined with aluminium, calcium, magnesium, iron and titanium in various silicate minerals. Many other elements occur in smaller amounts, though the heavier ones are very rare.
The spatial distribution of these elements is uneven. On average, lunar highland rocks contain nearly three times as much aluminium, about one third as much iron, and less than one fifth as much titanium as maria basalts. But these figures still under-represent the moon’s diversity. Samples from different landing sites show significant variations, as do the lunar meteorites found on Earth.
Apollo 11, 12 and 14 all targeted lowland areas. Their samples differed noticeably from each other, and more markedly from material gathered on highland sites by Apollo 15 and 16. Analogous differences were found between lowland samples returned from Luna 16 and 24 and highland material provided by Luna 20. From the unusual terrain of Mare Serenitatis Apollo 17 brought unique finds which further enriched the picture. Later, a broader context for all these results emerged from lengthy spectroscopic surveys made by orbiting probes, including the US’s Lunar Prospector (1998–9) and the European Space Agency’s SMART-1 (2003–6).
Under a floating crust of lighter substances, denser materials like the iron-rich olivine sank into the still-molten magma beneath
Overall, the commonest lunar highland rock is anorthosite (which is also widespread on Earth). Lunar anorthosites consist mostly of plagioclase feldspar, a mixture whose principal component is anorthite (CaAl2Si2O8), plus some albite (NaAlSi3O8). Highland rocks may also include smaller quantities of other minerals – including olivine (a mixture of Mg2SiO4 and Fe2SiO4) and ilmenite (mainly FeTiO3), both of which are more plentiful in the maria.
Astronauts exploring the maria on foot found rock fragments lighter in colour than the surrounding material, and chemically different from it. They proved to be debris from highland feldspar rocks, shattered and scattered by massive meteoric impacts. Trapped argon bubbles in these specimens (produced by radioactive decay of potassium-40) revealed that they had solidified much earlier than the maria basalt beneath them.
Isotopic dating of its oldest rocks indicates that the lunar surface began solidifying about 4.5 billion years ago. Under a floating crust of lighter substances (mainly anorthosites), denser materials like the iron-rich olivine sank into the still-molten magma beneath. Meanwhile, collisions with asteroid-sized bodies made huge depressions in the surface, some of which became filled with molten rock to create the maria. This process, however, was more complex than it first seemed.
The relative ages of maria can be estimated from the extent to which their (originally smooth) surfaces have been cratered by subsequent impacts, but isotopic dating of samples can show more precisely when any particular surface solidified. It appears that most of the basins created by asteroid impacts were not filled with magma immediately – indeed, the delay could last many millions of years. This upwelling therefore required another contributory factor, which further chemical investigations revealed.
Kreeping towards understanding
Many Apollo samples included an unexpected component – a mixture known as ‘Kreep’, containing potassium (K), rare earth elements (REE) and phosphorus (P). Due to various physical and chemical constraints, these elements were reluctant to crystallise with the substances surrounding them, and so became concentrated in the remaining liquid magma. When they did eventually solidify, they were often accompanied by other relatively unsociable elements. These included uranium and thorium which – along with the radioactive potassium-40 – could generate enough heat to melt rocks.
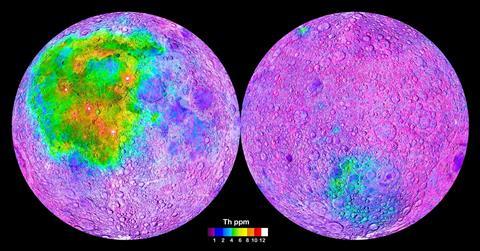
It seems probable that sub-surface concentrations of these heat-producing elements powered the volcanic activity which penetrated the lunar crust and filled the impact depressions. This thesis is corroborated by a surprising anomaly. The side of the moon facing away from the Earth has many impact basins, but hardly any maria. This is attributed to resistance from the far side’s crust, which satellite surveys have shown to be about 15km thicker than the crust on the side facing us. Also significant is the relative scarcity – as revealed by satellite observations – of heat-generating Kreep deposits on the far side.
As evidence about the moon’s composition accumulated, debate about its origins intensified. Some astronomers had suggested that the moon was spun off from the proto-Earth – but computer modelling shows this would require an improbably high rotational velocity. Others thought the moon coalesced from a cloud of dust orbiting the Earth – though how this cloud originated remained mysterious. And some assumed that the Earth captured a fully-formed moon which crossed its path during the solar system’s chaotic youth. Computer models, however, indicate that unless the trajectories of two such bodies were optimally aligned, either a miss or a collision would have resulted.
At present, the most persuasive origin theory is that collision with a Mars-sized body remodelled the Earth, and created the moon. Strong evidence for this comes from the remarkable similarities found between lunar and terrestrial chemistry. Although the moon is poorer in heavier elements, the relative proportions of its lighter elements are very similar to those on Earth. Moreover, the isotopes of certain elements (notably oxygen) occur in near-identical proportions in terrestrial and lunar rocks. In contrast, other extra-terrestrial samples – from meteorites, and from the Mars probes – exhibit much greater chemical and isotopic diversity.

It therefore seems likely that about 4.6 billion years ago a stray planet (retrospectively named Theia) collided with the Earth so violently that almost all of its matter – and much of the Earth’s – vaporised. This incandescent gas became thoroughly mixed before most of it re-solidified around the Earth’s core, while the remainder condensed to form the moon.
Applying new techniques to existing resources can still tell us more about the Moon’s composition and history – this year, the Apollo Next Generation Sample Analysis programme committed $8 million (£6.3 million) to fresh projects – but a new surge of exploration has already begun. China’s Chang’e 4 lunar probe is now transmitting exciting data from the Aitken Basin, near the moon’s south pole. This huge depression – 134km wide and 6km deep – was probably created by a 200km wide asteroid around 4 billion years ago. Scientists have long believed that it cracked the lunar crust, releasing molten material from the mantle (though not enough of it to form very large dark areas like the maria on the moon’s near side).
Spectrochemical analyses by Chang’e 4 have now identified surface minerals which support this assertion, and more results are expected shortly. Meanwhile Nasa is urgently seeking Federal funding for human landings by 2024, and entrepreneurs are preparing privately funded missions to search for exploitable mineral resources. Much has already been learned about the moon’s chemistry, but further surprises may still await the next generation of explorers.
Mike Sutton is a science historian based in Newcastle, UK
Further reading
B L Joliffe, M A Wieczorek, C K Shearer and C R Neal (eds), New Views of the Moon, de Gruyter & Co, 2018
D Whitehouse, The Moon: A Biography, Headline, 2001













No comments yet